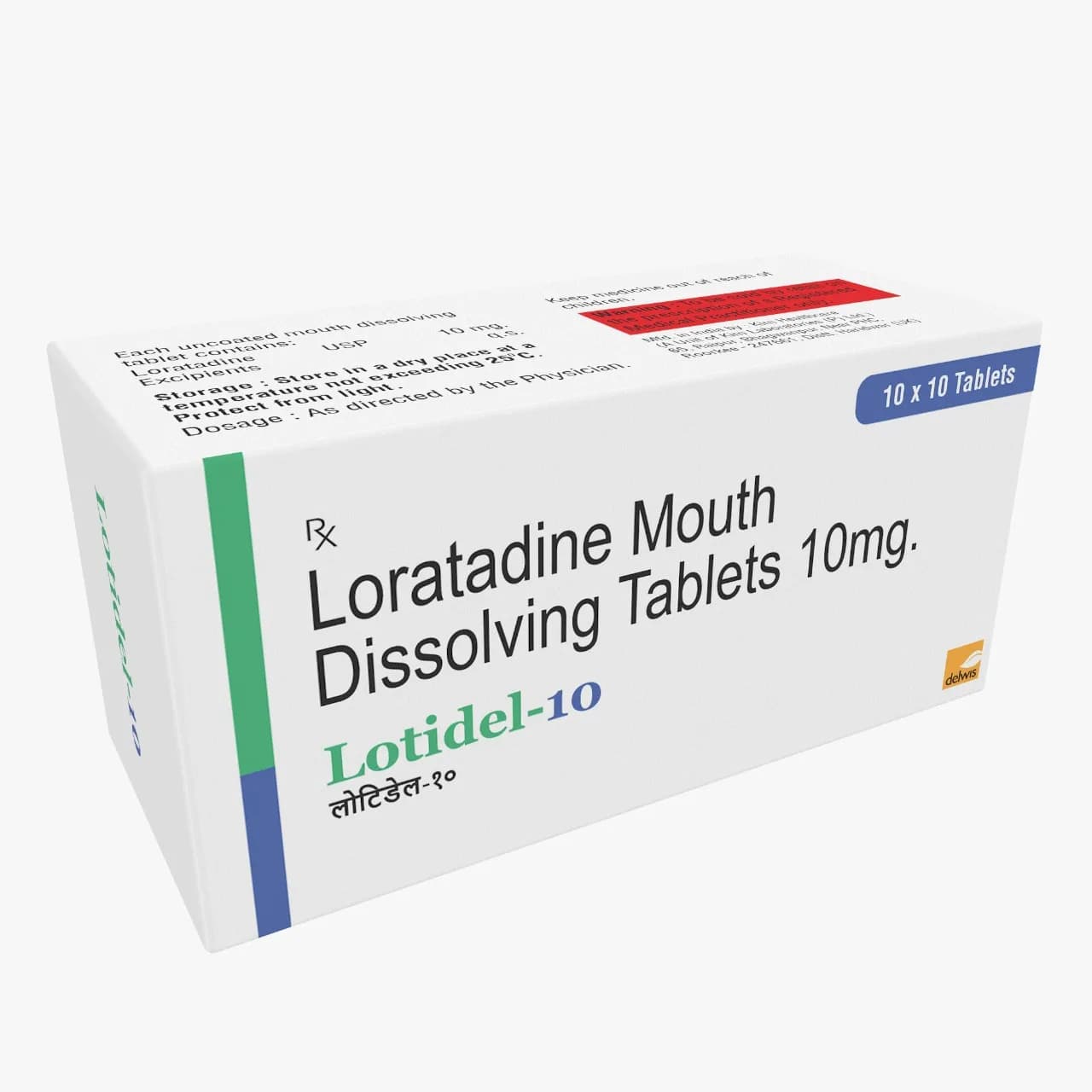
Lotidel 10 is a mouth-dissolving tablet formulated with loratidine 10mg as the active ingredient, combined with excipients to ensure optimal dissolution and stability. The formulation leverages advanced tablet technology to enable rapid disintegration in the oral cavity, eliminating the need for water and improving patient compliance. The manufacturing process involves precise compression of the active pharmaceutical ingredient (API) with excipients, followed by drying and coating to ensure uniformity and stability. The mouth-dissolving technology requires specialized equipment and process control to maintain tablet integrity while ensuring consistent drug release. Key formulation parameters include disintegration time, dissolution rate, and compatibility of excipients with the API. The product’s design supports scalability and meets pharmaceutical standards for solid dosage forms, making it suitable for diverse therapeutic applications.
Manufacturing Capability
Our pharmaceutical manufacturing infrastructure is equipped with state-of-the-art facilities, including high-speed tablet presses, drying chambers, and coating systems, enabling efficient production of mouth-dissolving tablets. The facility adheres to Good Manufacturing Practice (GMP) standards, ensuring compliance with regulatory requirements for pharmaceutical quality. Sterile processing capabilities are available for critical dosage forms, with aseptic filling lines to prevent contamination. The production process is designed for scalability, allowing seamless transitions from small-scale development to large-volume manufacturing. Quality systems include real-time monitoring, automated data collection, and validated cleaning protocols to maintain product consistency. Our facility is certified under ISO 9001 and ISO 14001, reflecting commitment to quality and environmental sustainability.
Available Strengths and Packaging
Composition: Loratidine 10mg Tab Mouth Dissolving
Packaging: 10x10 Alu Alu
Lotidel 10 is available in a single strength of 10mg, formulated as a mouth-dissolving tablet. The packaging consists of 10x10 aluminum blister packs, ensuring tamper-evident protection and extended shelf life. The Alu Alu packaging is designed for stability under varied storage conditions, complying with pharmaceutical packaging standards for solid dosage forms.
Contract Manufacturing / Third Party Manufacturing
As a trusted manufacturer, we offer comprehensive contract manufacturing and third-party production services tailored to pharmaceutical buyers. Our capabilities include private label manufacturing, where we produce branded dosage forms under a client’s proprietary name, as well as contract manufacturing for generic and branded products. We support customization of packaging, dosage strengths, and formulation specifications to meet specific market demands. Our flexible production model accommodates both small and large-scale orders, ensuring timely delivery without compromising quality. For pharmaceutical exporters and distributors, we provide end-to-end manufacturing solutions, from API sourcing to finished product packaging, ensuring compliance with global regulatory frameworks.
Quality Assurance
Our quality assurance systems are built on rigorous analytical validation, batch testing, and adherence to pharmaceutical standards. Each production batch undergoes comprehensive testing, including HPLC analysis for API content, dissolution testing to confirm drug release profiles, and physical testing for tablet hardness and disintegration time. Quality control measures are integrated throughout the manufacturing process, with real-time monitoring of critical parameters such as temperature, humidity, and equipment calibration. We maintain detailed batch records and documentation to ensure traceability and compliance with regulatory requirements. Our laboratory is equipped with advanced analytical instruments, including UV-Vis spectrophotometers and moisture analyzers, to validate product consistency and purity.
Regulatory Compliance
Lotidel 10 complies with Good Manufacturing Practice (GMP) standards set by the FDA, EMA, and WHO, ensuring adherence to international pharmaceutical regulations. Our manufacturing processes are validated to meet the requirements of the United States Pharmacopeia (USP) and European Pharmacopeia (EP), guaranteeing product quality and safety. The facility is certified under ISO 9001 for quality management and ISO 14001 for environmental management, reflecting our commitment to sustainable practices. Additionally, we maintain compliance with the ICH Q7 guidelines for API manufacturing and adhere to the WHO’s guidelines for dosage form stability and packaging. These certifications enable seamless export to global markets, including the EU, US, and Asia-Pacific regions.
Global Supply Capability
We provide robust global supply capabilities, including export to over 50 countries and a well-established international distribution network. Our bulk supply capacity supports large-volume orders, with inventory management systems ensuring timely delivery and minimal lead times. The supply chain is designed for reliability, with partnerships for logistics and customs clearance to facilitate smooth cross-border shipments. We offer flexible packaging options and compliance documentation to meet the requirements of international regulatory bodies. Our export capabilities are supported by a dedicated team specializing in global trade compliance, ensuring adherence to import regulations and phytosanitary standards.
Why Choose Us
Pharmaceutical distributors, exporters, and contract manufacturing partners benefit from our expertise in advanced dosage form development, regulatory compliance, and scalable production. Our commitment to quality, combined with a proven track record in manufacturing mouth-dissolving tablets, ensures reliable supply chains and product consistency. As a certified manufacturer, we offer competitive pricing, rapid turnaround times, and customized solutions tailored to market-specific needs. Our global supply network and compliance certifications enable seamless entry into international markets, while our dedicated customer support team ensures end-to-end satisfaction.
Bulk Supply Inquiry
For bulk supply inquiries, contact our sales team at info@delwishealthcare.com.
FAQs
What manufacturing processes are used for Lotidel 10?
Lotidel 10 is manufactured using advanced tablet compression and drying techniques, with specialized equipment to ensure consistent disintegration and dissolution.
Can Lotidel 10 be produced in different packaging formats?
While the standard packaging is 10x10 Alu Alu, we offer customization for alternative packaging formats upon request.
What regulatory certifications does Lotidel 10 hold?
Lotidel 10 complies with GMP, WHO, FDA, and EMA guidelines, with certifications under ISO 9001 and ISO 14001.
Is bulk supply available for Lotidel 10?
Yes, we provide bulk supply options with flexible order quantities to meet large-scale distribution needs.
What quality control measures are in place for Lotidel 10?
Each batch undergoes rigorous testing, including HPLC analysis, dissolution testing, and physical property validation.
Can Lotidel 10 be exported to international markets?
Yes, our compliance with global regulatory standards enables seamless export to over 50 countries.
What is the lead time for bulk orders?
Lead times vary based on order size, but we prioritize timely delivery with a minimum of 4–6 weeks for standard orders.
How does your company ensure supply chain reliability?
We maintain a robust logistics network and inventory management system to ensure consistent supply and minimize delays.
Can Lotidel 10 be customized for private label manufacturing?
Yes, we offer private label manufacturing services to meet specific branding and packaging requirements.
What documentation is provided for export compliance?
We supply complete regulatory documentation, including Certificates of Analysis (CoA), batch records, and compliance certificates for global markets.