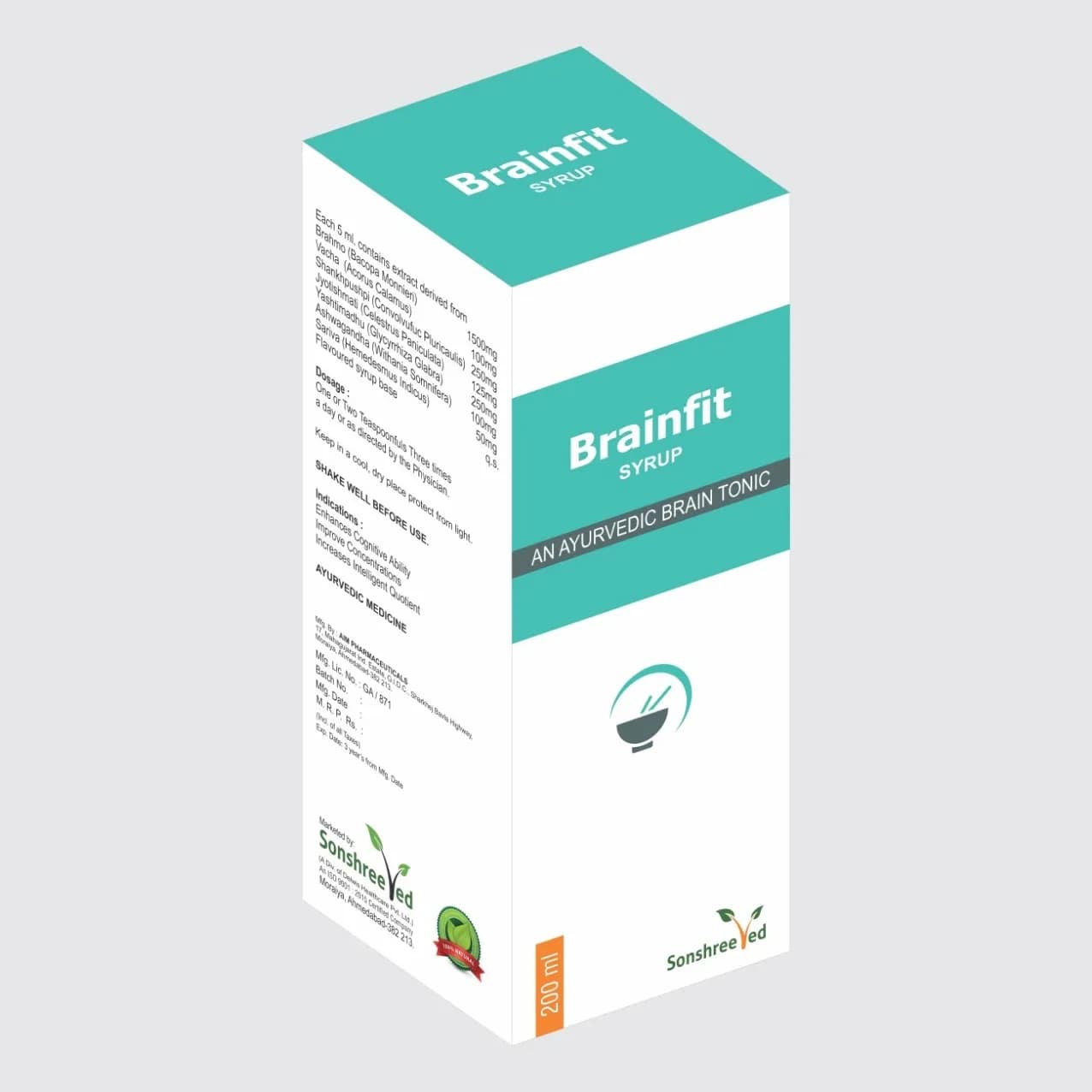
Brainfit Syp is a meticulously formulated herbal syrup designed to meet the stringent requirements of pharmaceutical manufacturing. The formulation integrates standardized extracts of Brahmi (Bacopa Monnieri), Vacha (Acorus Calamus), Shankhpushpi (Convolvulus pluricaulis), Jyotishmati (Celestus paniculata), Yashtimadhu (Glycyrrhiza glabra), Ashwagandha (Withania somnifera), and Sariva (Hemidesmus indicus), each selected for its pharmacological properties and synergistic potential. The syrup’s composition is optimized for stability, solubility, and bioavailability, ensuring consistent quality and efficacy. The formulation employs a balanced combination of excipients, including purified water, preservatives, and flavoring agents, to maintain product integrity during storage and transportation. The manufacturing process prioritizes homogeneity, ensuring uniform distribution of active ingredients across each batch. The syrup’s viscosity and pH are carefully controlled to enhance patient compliance and shelf-life, while its transparent, amber-colored packaging safeguards against light degradation. From a formulation perspective, Brainfit Syp is engineered to meet global pharmaceutical standards, with a focus on scalability, reproducibility, and regulatory compliance.
Manufacturing Capability
Our state-of-the-art pharmaceutical manufacturing infrastructure is designed to produce Brainfit Syp with precision and consistency. The facility adheres to Good Manufacturing Practice (GMP) guidelines, ensuring compliance with international quality standards. The production process includes advanced sterile processing capabilities, including HEPA-filtered environments and validated aseptic techniques, to prevent contamination. Automated blending and homogenization systems ensure uniform distribution of active ingredients, while precision dosing equipment maintains strict adherence to formulation specifications. The facility is equipped with ISO 9001-certified quality management systems, enabling full traceability of raw materials and finished products. Scalability is a core strength, with the ability to produce bulk quantities of Brainfit Syp while maintaining product consistency. The manufacturing line supports flexible production schedules, accommodating both small-scale trials and large-volume commercial orders. Rigorous validation protocols, including process analytical technology (PAT) and in-process quality control, ensure that every batch meets predefined specifications.
Available Strengths and Packaging
Brainfit Syp is formulated with standardized herbal extracts in fixed concentrations: Brahmi (1500mg), Vacha (100mg), Shankhpushpi (100mg), Jyotishmati (125mg), Yashtimadhu (250mg), Ashwagandha (100mg), and Sariva (50mg) per 200ml volume. The product is packaged in a 200ml bottle with an outer protective layer, ensuring safe storage and transportation. This packaging design provides resistance to physical damage and environmental factors, maintaining product stability during distribution.
Contract Manufacturing / Third Party Manufacturing
As a trusted pharmaceutical manufacturer, we offer comprehensive contract manufacturing and third-party production services tailored to meet the needs of global buyers. Our capabilities include private-label manufacturing, where we produce Brainfit Syp under a client’s brand name, ensuring full customization of packaging, labeling, and quality control protocols. We also provide flexible contract manufacturing options, enabling pharmaceutical companies to outsource production while retaining control over specifications and regulatory compliance. Our facility supports both small-scale development and large-volume commercial production, with the ability to scale operations to meet market demands. We prioritize transparency, offering detailed documentation, batch-specific reports, and compliance certifications to ensure seamless integration into a buyer’s supply chain.
Quality Assurance
Quality assurance is central to our manufacturing process for Brainfit Syp. Every batch undergoes rigorous testing at multiple stages, including raw material verification, in-process controls, and final product validation. Analytical methods such as high-performance liquid chromatography (HPLC) and ultraviolet (UV) spectroscopy are employed to confirm the potency and purity of active ingredients. Batch testing ensures compliance with pharmacopeial standards, while microbial and physical tests confirm safety and stability. Our quality control systems are aligned with ISO 27001 and GMP guidelines, with continuous monitoring of critical parameters such as pH, viscosity, and microbial load. All documentation is traceable and compliant with regulatory requirements, providing pharmaceutical buyers with confidence in product consistency and reliability.
Regulatory Compliance
Brainfit Syp is manufactured in full compliance with global regulatory frameworks, including Good Manufacturing Practice (GMP), World Health Organization (WHO) guidelines, and ISO 9001 quality management standards. Our facility is certified by regulatory authorities in multiple jurisdictions, ensuring adherence to international quality and safety benchmarks. The product meets pharmacopeial standards for active ingredients, excipients, and packaging materials, with documentation available for regulatory submissions. We maintain compliance with the U.S. FDA, EU GMP, and ICH guidelines, enabling seamless export to global markets. Our quality assurance team ensures that all documentation, including batch records, stability reports, and certificates of analysis (CoA), meets the requirements of pharmaceutical buyers and regulatory bodies.
Global Supply Capability
Our pharmaceutical manufacturing capabilities extend to global supply chains, with a focus on reliable, bulk supply solutions for international markets. We maintain an extensive distribution network, enabling efficient export to regions across Asia, Europe, Africa, and the Americas. The facility is equipped to handle large-volume orders, with a production capacity exceeding 1 million units per month. Our supply chain is designed for resilience, incorporating redundancy in logistics and real-time tracking systems to ensure timely delivery. Bulk supply options are tailored to meet the specific needs of buyers, with flexible packaging solutions and compliance certifications for international trade. We prioritize supply chain reliability, ensuring consistent delivery of high-quality Brainfit Syp to meet the demands of pharmaceutical distributors and exporters.
Why Choose Us
Pharmaceutical distributors, exporters, and contract manufacturing partners select us for our expertise in herbal formulation, compliance-driven manufacturing, and global supply capabilities. Our commitment to GMP standards, rigorous quality control, and scalable production ensures that Brainfit Syp meets the demands of diverse markets. As a trusted supplier, we offer competitive pricing, transparent documentation, and customized solutions to support your business objectives. Our facility’s compliance with WHO, FDA, and ISO standards simplifies regulatory submissions, while our flexible manufacturing options allow for rapid adaptation to market needs. By partnering with us, you gain access to a reliable, high-quality product with a proven track record in the pharmaceutical industry.
Bulk Supply Inquiry
For bulk supply inquiries, contact our team at info@delwishealthcare.com to discuss customized manufacturing, packaging, and distribution solutions tailored to your requirements.
FAQs
What is the manufacturing process for Brainfit Syp?
Brainfit Syp is produced using GMP-compliant manufacturing techniques, including automated blending, homogenization, and aseptic processing to ensure product consistency and safety.
Can Brainfit Syp be customized for private labeling?
Yes, we offer private-label manufacturing services, allowing clients to brand Brainfit Syp under their own label while maintaining quality and compliance standards.
What certifications does your facility hold?
Our facility is certified under GMP, ISO 9001, and WHO guidelines, ensuring adherence to international pharmaceutical quality standards.
Is Brainfit Syp suitable for export to international markets?
Yes, the product is formulated and manufactured to meet the regulatory requirements of global markets, including the FDA, EU GMP, and ICH guidelines.
What is the shelf life of Brainfit Syp?
The product has a shelf life of 24 months when stored under recommended conditions, with stability testing confirming its efficacy and safety over time.
Can you accommodate large-scale bulk production?
Yes, our facility is designed for scalable production, with the capacity to meet high-volume orders while maintaining product quality and consistency.
What quality control measures are in place for Brainfit Syp?
Each batch undergoes rigorous testing, including HPLC, UV spectroscopy, microbial analysis, and physical testing, to ensure compliance with pharmacopeial standards.
How do you ensure supply chain reliability?
We maintain a robust supply chain with real-time tracking, redundant logistics, and compliance certifications to ensure timely and reliable delivery of bulk orders.