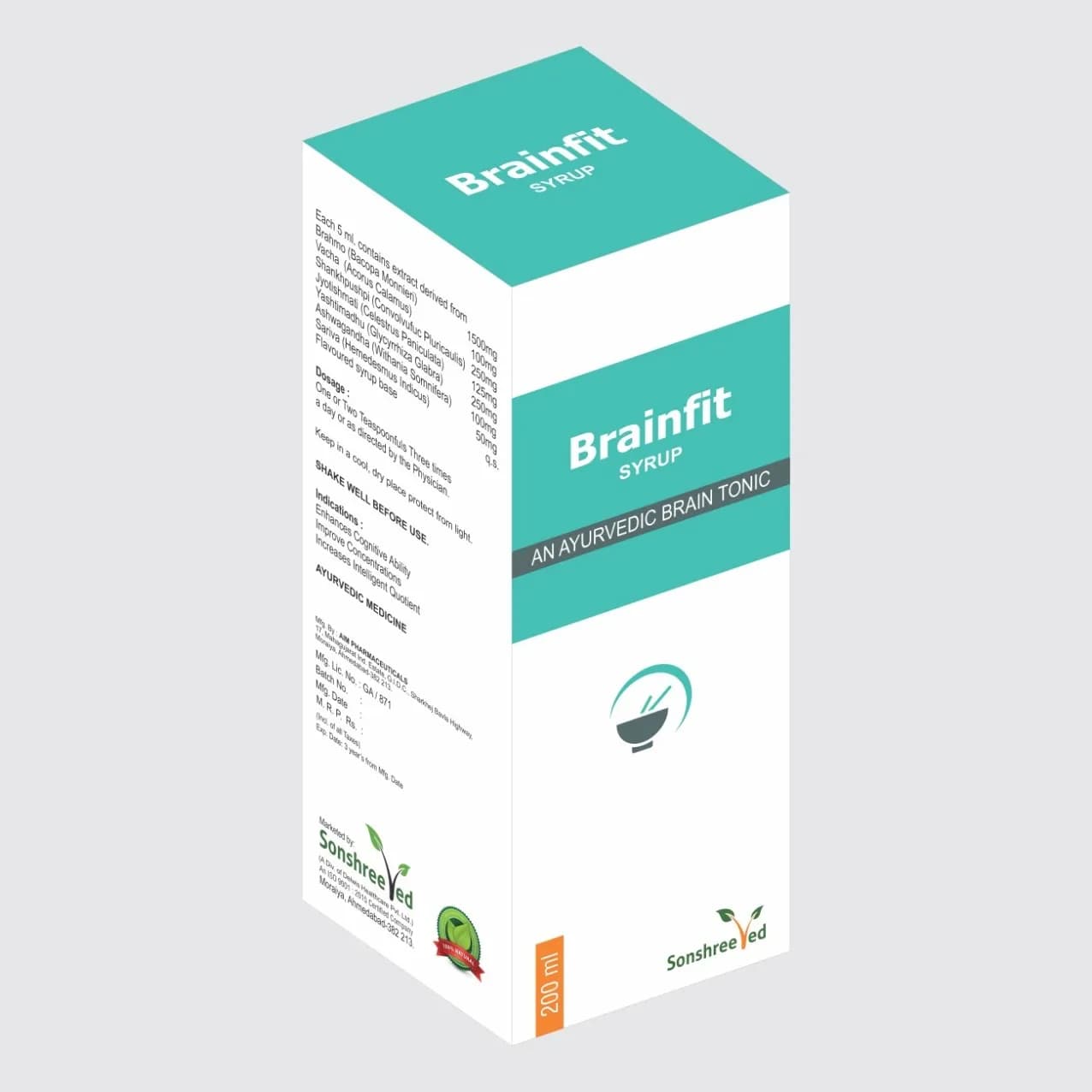
Ayuritis is a meticulously formulated capsule designed to address musculoskeletal discomfort, incorporating a balanced blend of active pharmaceutical ingredients (APIs) and excipients to ensure stability, bioavailability, and compliance with pharmaceutical standards. The formulation leverages advanced excipient technology to enhance dissolution rates and maintain structural integrity during storage. Key components include a synergistic combination of anti-inflammatory and analgesic agents, encapsulated in a robust, moisture-resistant capsule shell to preserve potency. The product’s pharmaceutical characteristics are optimized for uniformity, disintegration, and dissolution, ensuring consistent performance across batches. Manufacturing processes prioritize precision in blending, compression, and coating to meet stringent quality requirements. The formulation is designed for scalability, enabling efficient production while maintaining adherence to regulatory guidelines for pharmaceutical excipients and API compatibility.
Manufacturing Capability
Our pharmaceutical manufacturing infrastructure is equipped with state-of-the-art facilities compliant with current Good Manufacturing Practices (cGMP) and ISO 9001 standards. The production line includes automated blending, high-speed tablet compression, and advanced capsule filling systems to ensure precision and consistency. Sterile processing capabilities are available for critical components, supported by ISO Class 7 and Class 8 cleanroom environments. The facility adheres to rigorous quality control protocols, including real-time monitoring of critical process parameters such as temperature, humidity, and particle size distribution. Production scalability is achieved through modular design, enabling seamless transitions from small-scale trials to large-volume manufacturing. Quality systems encompass comprehensive documentation, traceability, and validation of all processes, ensuring compliance with global pharmaceutical standards.
Available Strengths and Packaging
Ayuritis is formulated in standard strengths tailored to address musculoskeletal conditions effectively, with dosage levels calibrated for therapeutic efficacy and safety. The product is available in the packaging format of 6x10 Alu Alu, ensuring secure, tamper-evident storage and transport. This packaging design optimizes shelf life while meeting regulatory requirements for pharmaceutical product integrity.
Contract Manufacturing / Third Party Manufacturing
As a trusted manufacturer, we offer comprehensive contract manufacturing, private label production, and third-party manufacturing services to meet the diverse needs of pharmaceutical buyers. Our capabilities include full-scale production of API-based formulations, excipient blending, and finished dosage form manufacturing, with flexibility to accommodate custom specifications. We support private label manufacturing by providing end-to-end solutions, from formulation development to packaging and labeling. Third-party manufacturing partnerships are facilitated through strict confidentiality agreements and adherence to cGMP standards, ensuring seamless integration with client supply chains. Our services are ideal for pharmaceutical companies seeking to outsource production while maintaining control over quality and compliance.
Quality Assurance
Our quality assurance framework is built on rigorous analytical validation, batch testing, and adherence to pharmaceutical quality standards. Every production batch undergoes comprehensive testing, including high-performance liquid chromatography (HPLC), dissolution testing, and microbial limit checks to ensure product purity and potency. Quality control systems are integrated with advanced data analytics to monitor process variability and ensure consistency. Analytical validation protocols comply with ICH guidelines, while stability testing ensures shelf-life accuracy. All documentation is maintained in accordance with FDA and EMA requirements, providing full traceability and audit readiness.
Regulatory Compliance
Ayuritis is manufactured in full compliance with global regulatory frameworks, including cGMP, WHO guidelines, and ISO 9001 standards. The product adheres to FDA, EMA, and USP monograph requirements, ensuring eligibility for market approval in major pharmaceutical markets. Our facility is certified under ISO 17025 for testing and calibration, and all documentation meets the stringent requirements of the International Council for Harmonisation (ICH). Compliance with WHO prequalification standards ensures accessibility in low- and middle-income countries. Regulatory submissions are supported by detailed dossiers, including process validation, stability data, and safety profiles, enabling swift approval processes.
Global Supply Capability
Our global supply chain is designed to deliver reliable, high-quality pharmaceutical products to international markets. With a robust network of distributors and logistics partners, we ensure timely delivery of bulk supplies to over 50 countries. The 6x10 Alu Alu packaging format is optimized for efficient storage and transport, reducing logistical complexities. Bulk supply capacity is supported by scalable production capabilities, enabling rapid response to market demands. Our supply chain reliability is reinforced by real-time tracking systems, inventory management, and contingency planning to mitigate disruptions. Export capabilities are further enhanced by compliance with international shipping regulations and phytosanitary requirements.
Why Choose Us
Pharmaceutical distributors, exporters, and contract manufacturing partners benefit from our expertise in quality-driven manufacturing, regulatory compliance, and global supply chain management. Our commitment to cGMP standards, combined with flexible production capabilities, ensures that clients receive products meeting the highest industry benchmarks. As a trusted supplier, we offer tailored solutions to address unique market needs, from formulation development to packaging and distribution. Our focus on innovation, transparency, and sustainability positions us as a reliable partner for long-term collaboration.
Bulk Supply Inquiry
We provide seamless bulk supply solutions for pharmaceutical buyers. For inquiries regarding large-scale orders, packaging specifications, or regulatory documentation, contact our team at info@delwishealthcare.com.
FAQs
What are the available strengths for Ayuritis?
Ayuritis is formulated in standard strengths optimized for therapeutic efficacy, with dosage levels calibrated to ensure consistent performance.
What packaging options are available for Ayuritis?
The product is available in 6x10 Alu Alu packaging, designed for secure storage and efficient distribution.
Does Ayuritis comply with global regulatory standards?
Yes, Ayuritis is manufactured in compliance with cGMP, WHO guidelines, ISO 9001, and regional regulatory frameworks such as FDA and EMA.
Can Ayuritis be produced in custom strengths?
Yes, our contract manufacturing services support custom strength formulations to meet specific therapeutic requirements.
What quality control measures are in place for Ayuritis?
Every batch undergoes rigorous testing, including HPLC, dissolution, and microbial limit checks, ensuring compliance with pharmaceutical standards.
Is Ayuritis suitable for export to international markets?
Yes, the product is designed for global distribution, with packaging and documentation compliant with international shipping and regulatory requirements.
What is the minimum order quantity for bulk supply?
Bulk supply options are flexible, with quantities tailored to meet client needs. Contact us for customized solutions.
How does your manufacturing process ensure product consistency?
Our automated production systems and real-time monitoring ensure uniformity, with strict validation protocols to maintain batch consistency.
Can Ayuritis be produced under private label manufacturing?
Yes, we offer private label manufacturing services, allowing clients to brand the product while maintaining quality and compliance.
What is the shelf life of Ayuritis?
The shelf life is determined through stability testing and is clearly indicated on the packaging, ensuring product potency until the expiration date.