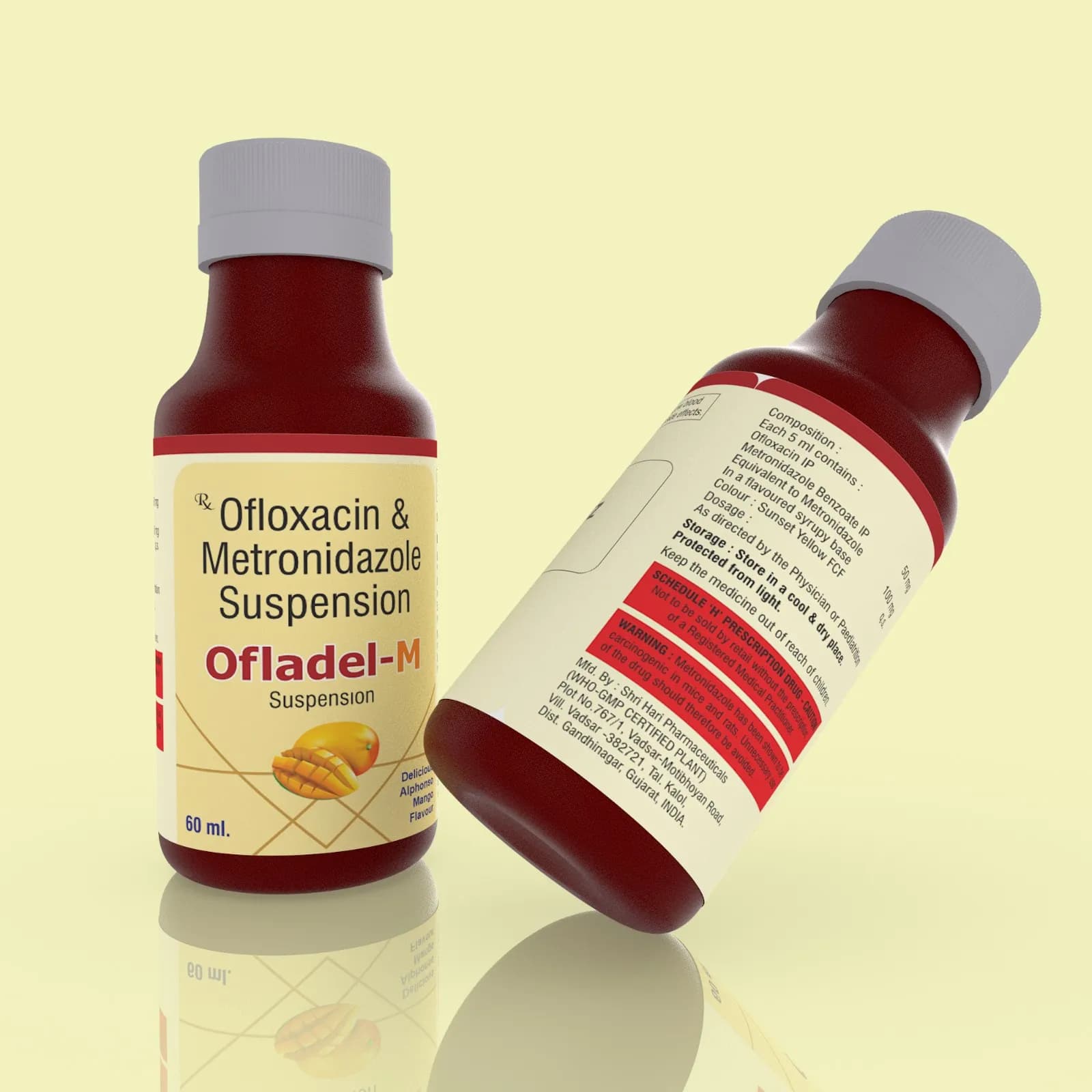
Ofladel - M Susp is a dual-action antibiotic suspension designed for pharmaceutical applications, combining Ofloxacin (a fluoroquinolone) and Metronidazole (an antiprotozoal and antibacterial agent). The formulation ensures optimal solubility and stability of both active ingredients in a suspension medium, enabling uniform dispersion for consistent dosing. The suspension’s pH and viscosity are carefully balanced to enhance patient compliance while maintaining the chemical integrity of the active pharmaceutical ingredients (APIs). The product’s formulation adheres to pharmaceutical standards for oral suspensions, ensuring compatibility with pediatric and adult populations requiring liquid dosage forms. Excipients such as flavoring agents, preservatives, and thickening agents are incorporated to improve sensory attributes and shelf life without compromising therapeutic efficacy. The combination of Ofloxacin and Metronidazole provides synergistic antimicrobial activity, making it suitable for treating mixed infections requiring broad-spectrum coverage.
Manufacturing Capability
Our pharmaceutical manufacturing infrastructure is designed to meet the stringent demands of producing high-quality antibiotic suspensions like Ofladel - M Susp. The facility is equipped with state-of-the-art aseptic processing lines, enabling sterile filling of suspensions in compliance with GMP standards. Advanced mixing and homogenization systems ensure uniform distribution of APIs, while temperature-controlled environments prevent degradation of sensitive compounds. The production process adheres to validated protocols for particle size reduction, suspension stability testing, and microbial contamination control. Scalability is achieved through modular production lines capable of handling bulk orders while maintaining consistency in quality and potency. Our quality systems include real-time monitoring, traceability mechanisms, and rigorous documentation to ensure compliance with global regulatory frameworks.
Available Strengths and Packaging
Composition: Ofloxacin 50mg + Metronidazole 100mg Suspension
Packaging: 60ml
Ofladel - M Susp is available in a 60ml single-dose packaging format, ensuring convenient administration and accurate dosing. The formulation is designed to deliver 50mg Ofloxacin and 100mg Metronidazole per 5ml of suspension, providing precise therapeutic dosing. The packaging is optimized for stability, with airtight seals and light-protective layers to preserve the integrity of the active ingredients.
Contract Manufacturing / Third Party Manufacturing
As a trusted manufacturer, we offer comprehensive contract manufacturing and third-party production services tailored to pharmaceutical buyers. Our facility supports private label manufacturing, enabling clients to brand Ofladel - M Susp under their own trademarks while maintaining compliance with regulatory standards. We provide end-to-end solutions, from formulation development to packaging and labeling, ensuring flexibility in meeting diverse market requirements. Our team of experts collaborates closely with clients to align production processes with their specifications, including dosage form customization, packaging design, and regulatory documentation. This service is ideal for pharmaceutical companies seeking to expand their product portfolios without investing in in-house manufacturing capabilities.
Quality Assurance
Our quality assurance framework is built on rigorous analytical validation, batch testing, and adherence to pharmaceutical quality standards. Every batch of Ofladel - M Susp undergoes comprehensive testing for API content, pH, viscosity, and microbial contamination using validated methods such as HPLC, titration, and microbial limit testing. Stability studies are conducted to ensure product efficacy under varying storage conditions, while in-process controls monitor critical parameters during manufacturing. Our quality systems are aligned with USP, EP, and BP standards, ensuring consistency in potency, purity, and safety. Traceability is maintained through batch-specific documentation, enabling full transparency for regulatory audits and supply chain management.
Regulatory Compliance
Ofladel - M Susp is manufactured in full compliance with GMP, WHO guidelines, and ISO 9001:2015 standards. Our facility is certified by regulatory authorities in multiple jurisdictions, including the FDA, EMA, and PIC/S, ensuring adherence to global quality expectations. The product’s formulation and manufacturing processes are validated to meet the requirements of international pharmacopoeias, enabling seamless export to markets worldwide. We maintain detailed documentation for regulatory submissions, including stability data, analytical reports, and process validation records. Our commitment to compliance extends to environmental sustainability practices, ensuring ethical and responsible manufacturing.
Global Supply Capability
We specialize in exporting high-quality pharmaceutical products to global markets, with a robust international distribution network and bulk supply capacity. Our logistics partners facilitate reliable delivery to over 50 countries, ensuring timely and cost-effective supply chain solutions. The 60ml packaging format is optimized for both regional and international distribution, with compliance to IATA and IMDG regulations for safe transportation. Our bulk supply capabilities support large-scale orders, with production flexibility to meet varying demand. Supply chain reliability is ensured through redundant inventory management, real-time tracking systems, and partnerships with certified freight forwarders.
Why Choose Us
Pharmaceutical distributors, exporters, and contract manufacturing partners benefit from our expertise in producing high-quality antibiotic suspensions like Ofladel - M Susp. Our combination of GMP-certified manufacturing, regulatory compliance, and scalable production capabilities positions us as a reliable partner for global markets. We offer competitive pricing without compromising on quality, supported by a dedicated account management team to address client needs. Our commitment to innovation, sustainability, and customer-centric solutions ensures long-term partnerships and market success.
Bulk Supply Inquiry
For bulk supply inquiries, please contact us at info@delwishealthcare.com. Our team is ready to assist with order placement, customization, and regulatory documentation for seamless global distribution.
FAQs
What manufacturing standards does Ofladel - M Susp comply with?
Ofladel - M Susp is manufactured in compliance with GMP, WHO guidelines, and ISO 9001:2015 standards, ensuring adherence to global quality expectations.
Can Ofladel - M Susp be produced in different packaging formats?
Yes, our facility supports customization of packaging formats, including single-dose and bulk options, to meet specific market requirements.
What is the shelf life of Ofladel - M Susp?
The product has a shelf life of 24 months when stored under recommended conditions, as validated through stability studies.
Does your facility offer contract manufacturing services for similar products?
Yes, we provide end-to-end contract manufacturing services, including formulation development, packaging, and regulatory documentation.
How do you ensure the quality of API content in each batch?
Every batch undergoes rigorous HPLC and titration testing to confirm API content, with results documented for traceability.
Can Ofladel - M Susp be exported to international markets?
Yes, our compliance with global regulatory frameworks enables seamless export to over 50 countries.
What is the minimum order quantity for bulk supply?
We accommodate bulk orders starting from 1,000 units, with flexible production scaling to meet client needs.
How do you handle regulatory submissions for international markets?
Our team prepares comprehensive regulatory documents, including stability data and analytical reports, to facilitate approvals in target markets.
What quality control measures are in place for microbial contamination?
Microbial limit testing is conducted using validated methods to ensure the product is free from contaminants.
Can I request a sample of Ofladel - M Susp for evaluation?
Yes, we offer sample requests for evaluation, with details available upon contact with our sales team.