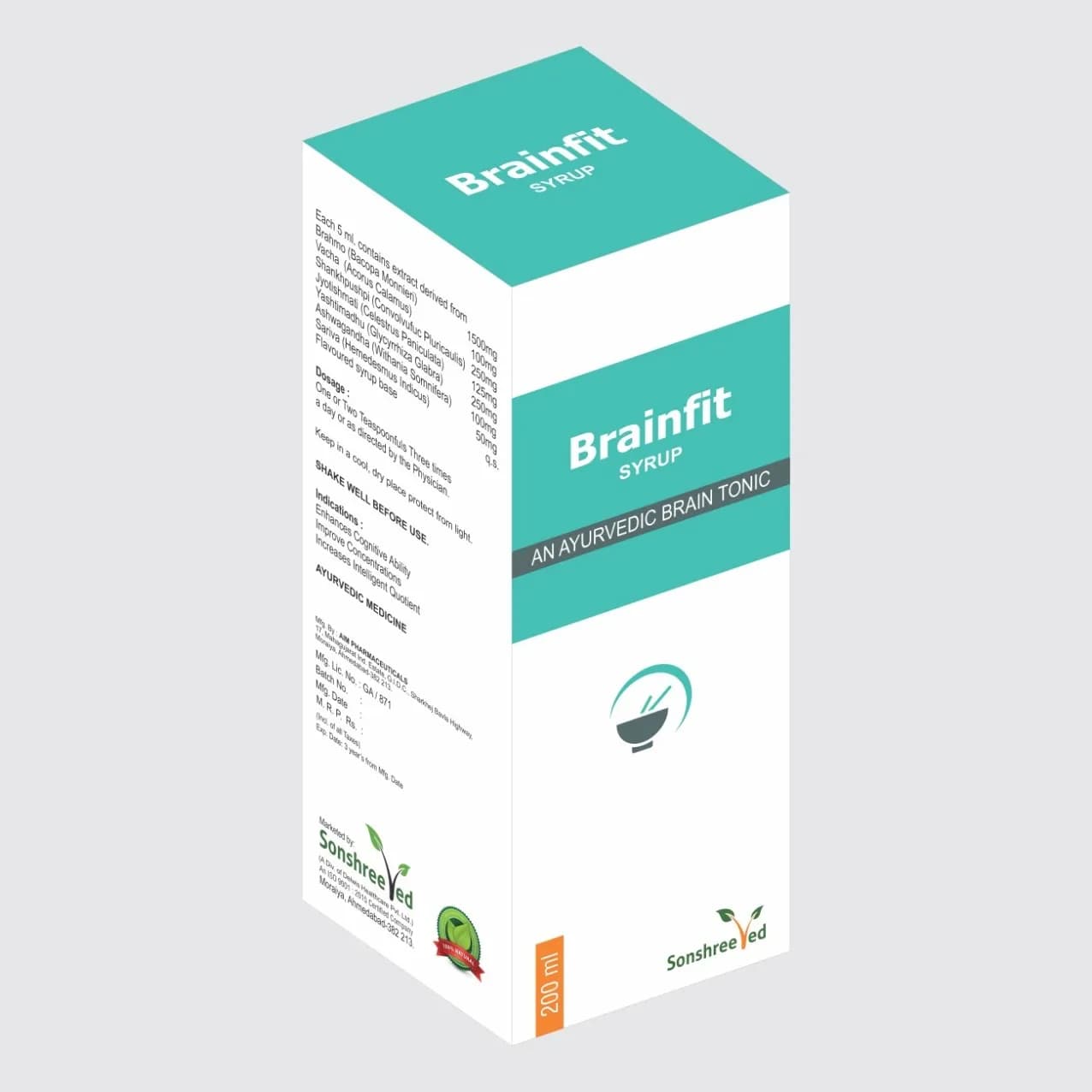
Fab Cap is a meticulously formulated weight loss capsule developed using advanced pharmaceutical techniques to ensure consistency, stability, and bioavailability of its herbal constituents. Each hard gelatin capsule contains a precise combination of standardized extracts, including Vrikshamla (Garcinia indica), Trikatu (a proprietary blend of Sundhi, Maricha, and Lindipiper), Haricha (Camellia sinensis), Shuddha Guggal (Commiphora mukul), Gudmar (Gymnema sylvestre), Methi (Trigonella foenum-graecum), Triphala (a blend of Amla, Harde, and Baheda), Karela (Momordica charantia), and Shuddha Shilajeet (Ashphaltum punjabianum). The formulation balances traditional Ayurvedic principles with modern pharmaceutical standards, ensuring uniformity in active ingredient concentration, dissolution rate, and capsule integrity. The use of hard gelatin capsules provides a stable matrix for the active compounds, while the inclusion of excipients ensures optimal flow, compression, and disintegration properties during manufacturing.
Manufacturing Capability
Our pharmaceutical manufacturing infrastructure is designed to meet the stringent demands of bulk production while maintaining compliance with global quality standards. The facility is equipped with state-of-the-art technology, including high-speed capsule filling machines, automated tablet compression systems, and advanced drying and cooling units to ensure consistent product quality. Sterile processing capabilities are available for sensitive formulations, with dedicated cleanrooms classified at ISO 7 and ISO 8 standards. All manufacturing processes adhere to Good Manufacturing Practice (GMP) guidelines, ensuring traceability, documentation, and rigorous quality control at every stage. The facility supports scalable production, with the ability to handle large-volume orders while maintaining precision in dosage uniformity and capsule integrity. Robust quality systems, including real-time monitoring and data integrity protocols, ensure compliance with regulatory requirements and minimize the risk of deviations during large-scale manufacturing.
Available Strengths and Packaging
Fab Cap is available in a single strength formulation, with each hard gelatin capsule containing the specified quantities of active ingredients as outlined in the composition. The product is packaged in 6x10 Alu Alu blister packs, ensuring secure, moisture-resistant storage and easy distribution for bulk supply. This packaging format is ideal for pharmaceutical buyers requiring efficient storage, transportation, and retail display solutions.
Contract Manufacturing / Third Party Manufacturing
As a trusted pharmaceutical manufacturer, we offer comprehensive contract manufacturing and third-party production services tailored to meet the needs of global buyers. Our capabilities include private label manufacturing, where clients can brand the product under their own name while leveraging our expertise in formulation, production, and quality assurance. We also provide flexible contract manufacturing options, enabling pharmaceutical companies to outsource entire production runs while retaining full control over specifications, packaging, and regulatory compliance. Our team works closely with clients to design custom formulations, optimize production timelines, and ensure adherence to international standards, making us a reliable partner for B2B buyers seeking scalable, high-quality solutions.
Quality Assurance
Our quality assurance framework is built on rigorous analytical validation, batch testing, and adherence to pharmaceutical quality standards. Every production batch undergoes comprehensive testing for purity, potency, and stability, including high-performance liquid chromatography (HPLC) and ultraviolet (UV) spectrophotometry to verify active ingredient content. We maintain detailed records of raw material sourcing, processing parameters, and finished product specifications to ensure traceability and transparency. In-line quality control measures, such as weight variation checks and disintegration testing, are performed during manufacturing to guarantee consistency. Additionally, our facility is certified under ISO 9001:2015, ensuring continuous improvement in quality management systems. These measures ensure that Fab Cap meets the highest standards of safety, efficacy, and regulatory compliance for global markets.
Regulatory Compliance
Fab Cap is manufactured in full compliance with Good Manufacturing Practice (GMP) guidelines, ensuring adherence to international pharmaceutical standards. The product meets the requirements of the World Health Organization (WHO) for herbal drug production, including documentation of raw material specifications, process validation, and finished product testing. Our facility is certified under ISO 9001:2015 and ISO 14001:2015 for quality management and environmental sustainability, respectively. Additionally, we maintain compliance with the United States Pharmacopeia (USP) and European Pharmacopoeia (Ph. Eur.) standards for active ingredient purity and formulation consistency. These certifications enable seamless export to markets across the globe, including the EU, North America, and Asia, while ensuring regulatory alignment with target regions.
Global Supply Capability
Our global supply capability is supported by an extensive international distribution network and robust logistics infrastructure, enabling efficient bulk supply to markets worldwide. We have established partnerships with trusted logistics providers to ensure timely delivery of products, with options for air, sea, and land transport tailored to specific regional requirements. Our facility is equipped to handle large-volume orders, with production capacity scalable to meet the demands of pharmaceutical exporters and distributors. We maintain a reliable supply chain through strategic inventory management, real-time tracking systems, and contingency planning to mitigate disruptions. This ensures consistent availability of Fab Cap for buyers seeking to meet market demand without compromising quality or compliance.
Why Choose Us
Pharmaceutical distributors, exporters, and contract manufacturing partners choose us for our expertise in herbal pharmaceuticals, compliance-driven manufacturing, and scalable production capabilities. Our commitment to quality, combined with a proven track record of regulatory compliance, ensures that Fab Cap meets the stringent requirements of global markets. We offer end-to-end solutions, from formulation development to bulk supply, enabling clients to streamline their operations and focus on market expansion. Our dedicated team provides personalized support, ensuring that each client’s needs are met with precision and efficiency. By partnering with us, buyers gain access to a reliable, high-quality product that aligns with international standards and supports their business objectives.
Bulk Supply Inquiry
For bulk supply inquiries, please contact our team at info@delwishealthcare.com. Our experts are ready to assist with order placement, customization, and logistics coordination to ensure seamless procurement and distribution.
FAQs
What certifications does Fab Cap hold?
Fab Cap is certified under GMP, ISO 9001:2015, and WHO guidelines, ensuring compliance with global pharmaceutical standards.
Can Fab Cap be customized for private labeling?
Yes, we offer private labeling services, allowing clients to brand the product under their own name while maintaining quality and compliance.
What is the shelf life of Fab Cap?
The product has a shelf life of 24 months when stored in its original packaging under controlled temperature and humidity conditions.
Is the packaging suitable for export?
The 6x10 Alu Alu blister pack is designed for export, offering moisture resistance and secure storage for international distribution.
Can you accommodate large-volume orders?
Yes, our scalable manufacturing infrastructure supports bulk production to meet the demands of global pharmaceutical buyers.
What quality control measures are in place?
Every batch undergoes rigorous testing for purity, potency, and stability, with documentation compliant with GMP and ISO standards.
Are there options for different packaging formats?
While the standard packaging is 6x10 Alu Alu, we can accommodate custom packaging requests upon consultation with our team.
How do you ensure regulatory compliance for international markets?
Our facility adheres to GMP, WHO, USP, and Ph. Eur. standards, enabling seamless compliance with regulatory requirements across global markets.
What is the minimum order quantity for bulk supply?
Minimum order quantities are flexible and can be discussed based on production capacity and client requirements.
How can I request a sample or quotation?
For samples or quotations, please contact us at info@delwishealthcare.com. Our team will provide detailed information and support.