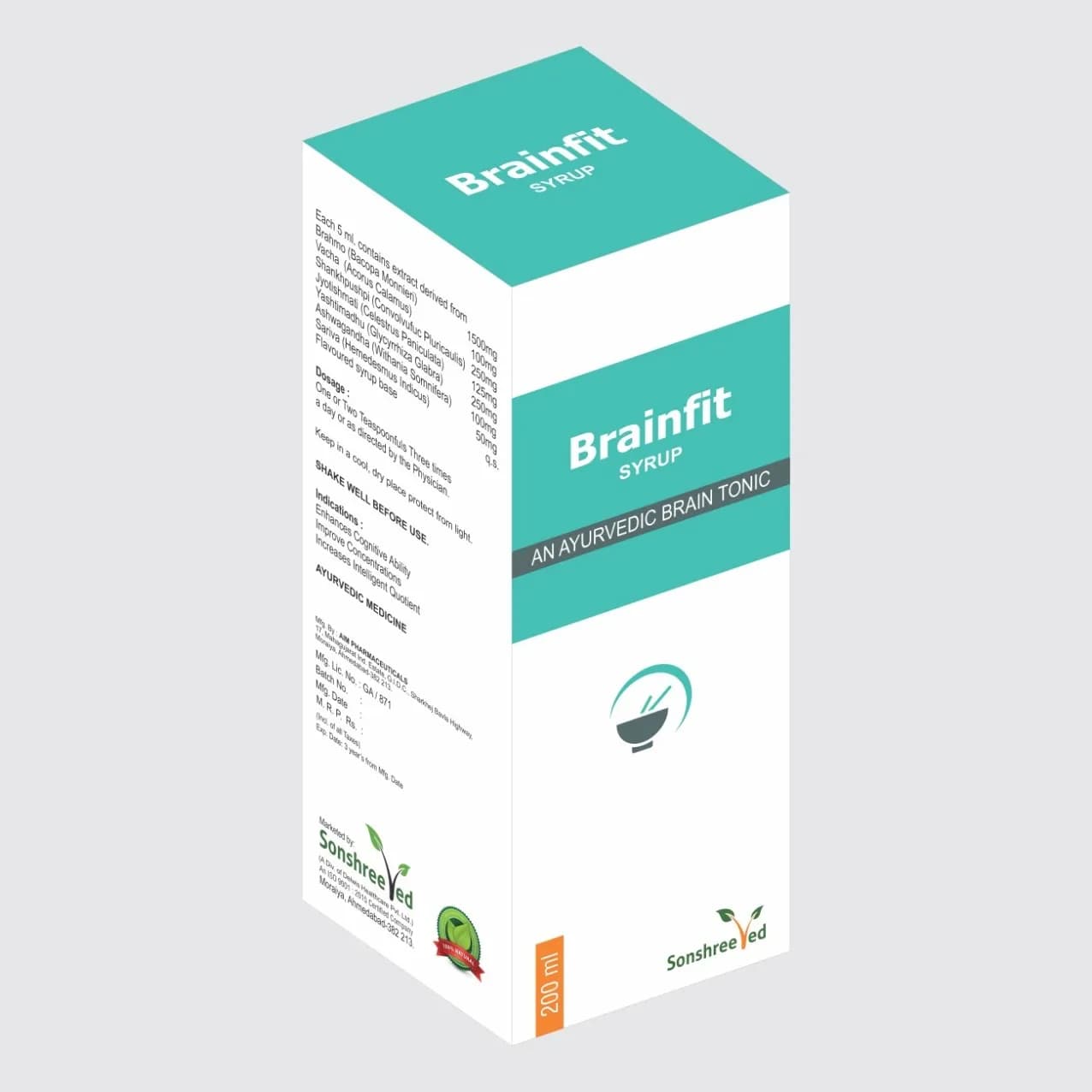
Jodayu Cap is a meticulously formulated capsule designed to address musculoskeletal discomfort, leveraging a combination of active pharmaceutical ingredients (APIs) and excipients to ensure stability, bioavailability, and therapeutic efficacy. The formulation incorporates a blend of analgesic, anti-inflammatory, and muscle-relaxant compounds, encapsulated in a controlled-release matrix to optimize absorption and prolong therapeutic effect. The capsule shell is composed of high-quality, pH-neutral materials, ensuring compatibility with gastric fluids and minimizing gastrointestinal irritation. The product’s formulation adheres to pharmaceutical-grade specifications, with excipients selected for their ability to enhance disintegration, flow properties, and tablet integrity. The manufacturing process involves precise blending, compression, and encapsulation techniques to maintain uniformity in dosage form, ensuring consistent performance across batches. The product’s stability profile is validated through accelerated degradation studies, confirming its shelf life under specified storage conditions.
Manufacturing Capability
Our manufacturing infrastructure is equipped with state-of-the-art facilities designed to produce pharmaceutical products like Jodayu Cap with precision and scalability. The facility features a fully integrated production line, including high-speed capsule filling machines, automated tablet compression systems, and advanced drying and packaging units. Sterile processing capabilities are maintained through ISO Class 7 cleanrooms, ensuring contamination-free environments for sensitive formulations. All manufacturing processes comply with current Good Manufacturing Practices (cGMP) and are validated to meet international pharmaceutical standards. The facility supports production scalability, enabling seamless transitions from small-scale trials to large-volume bulk supply. Quality systems are embedded throughout the manufacturing workflow, with real-time monitoring of critical parameters such as temperature, humidity, and particle size. Additionally, the facility is equipped with advanced analytical tools for in-process testing, ensuring adherence to specifications at every stage of production.
Available Strengths and Packaging
Jodayu Cap is formulated to deliver consistent therapeutic outcomes, with strengths tailored to meet clinical and market requirements. The product is available in standard dosage strengths, ensuring flexibility for diverse therapeutic applications. The packaging is provided in a 60 Cap Jar, designed for efficient storage, distribution, and retail display. The jar’s airtight closure and tamper-evident seal ensure product integrity and shelf-life compliance.
Contract Manufacturing / Third Party Manufacturing
As a leading manufacturer and supplier, we offer comprehensive contract manufacturing and third-party production services tailored to the needs of pharmaceutical buyers. Our capabilities include private label manufacturing, where we produce finished products under a client’s brand name, ensuring full customization of packaging, labeling, and formulation. We also provide contract manufacturing for bulk supply, accommodating both small and large-scale orders with strict adherence to quality and regulatory standards. Our team of experts works closely with clients to design formulations, optimize production processes, and ensure compliance with global regulatory frameworks. Whether you require a single product line or a multi-product portfolio, our flexible manufacturing infrastructure supports end-to-end solutions, from raw material sourcing to final packaging.
Quality Assurance
Quality assurance is a cornerstone of our pharmaceutical manufacturing process, ensuring that every batch of Jodayu Cap meets the highest standards of safety, purity, and efficacy. Our quality control systems include rigorous in-process testing, final product analysis, and stability studies to validate shelf-life and potency. Each batch undergoes analytical validation through high-performance liquid chromatography (HPLC), UV spectroscopy, and dissolution testing to confirm API uniformity and disintegration rates. We maintain detailed batch records and traceability systems to ensure transparency and compliance. Additionally, our facility is certified under ISO 9001:2015, reflecting our commitment to continuous improvement and customer satisfaction. All quality assurance protocols are aligned with international pharmacopoeial standards, including USP, EP, and BP, ensuring global market acceptance.
Regulatory Compliance
Our manufacturing processes are fully compliant with global regulatory frameworks, including current Good Manufacturing Practices (cGMP), World Health Organization (WHO) guidelines, and ISO 9001:2015 quality management standards. We adhere to stringent documentation requirements, ensuring that all production, testing, and quality control activities are traceable and auditable. Our facility is registered with regulatory authorities in key markets, including the FDA, EMA, and MHRA, enabling seamless export and market entry. We also maintain compliance with the ICH Q7 guidelines for active pharmaceutical ingredients and the ICH Q8 framework for pharmaceutical development. Our commitment to regulatory excellence ensures that Jodayu Cap meets the requirements of international buyers seeking reliable, compliant supply chains.
Global Supply Capability
As a trusted exporter and bulk supply provider, we have a robust international distribution network spanning major pharmaceutical markets. Our logistics infrastructure supports seamless export to over 50 countries, with partnerships that ensure timely delivery and compliance with import regulations. We offer bulk supply options tailored to meet the demands of large-scale distributors, wholesalers, and retail chains, with flexible order quantities and packaging solutions. Our supply chain is designed for reliability, featuring redundant inventory systems, real-time tracking, and contingency planning to mitigate disruptions. Whether you require small or large-volume orders, our team ensures that Jodayu Cap is delivered with precision, meeting deadlines and quality expectations.
Why Choose Us
Pharmaceutical distributors, exporters, and contract manufacturing partners choose us for our combination of technical expertise, regulatory compliance, and global reach. Our ability to deliver bulk supply with consistent quality and on-time delivery sets us apart in the competitive pharmaceutical landscape. As a manufacturer and supplier, we offer end-to-end solutions, from formulation development to market-ready products, ensuring that your business meets global demand. Our commitment to innovation, coupled with adherence to cGMP and ISO standards, guarantees that Jodayu Cap is a reliable choice for your portfolio. With a focus on sustainability and cost-efficiency, we provide value-driven solutions that align with your business objectives.
Bulk Supply Inquiry
We offer reliable bulk supply capabilities for Jodayu Cap, tailored to meet the needs of pharmaceutical buyers. For inquiries regarding large-volume orders, packaging customization, or export logistics, please contact us at info@delwishealthcare.com.
FAQs
What manufacturing standards does Jodayu Cap comply with?
Jodayu Cap is manufactured in compliance with cGMP, WHO guidelines, and ISO 9001:2015 standards, ensuring quality and safety for global markets.
Can Jodayu Cap be produced in different strengths?
Yes, our manufacturing capabilities allow customization of dosage strengths based on clinical and market requirements.
What is the shelf life of Jodayu Cap?
The product has a shelf life of 24 months when stored under recommended conditions (cool, dry, and protected from light).
Is Jodayu Cap suitable for export to international markets?
Yes, our regulatory compliance and global distribution network enable seamless export to over 50 countries.
What packaging options are available for Jodayu Cap?
The product is available in a 60 Cap Jar, designed for efficient storage and distribution.
Can you provide private label manufacturing services for Jodayu Cap?
Yes, we offer private label manufacturing, allowing you to brand the product under your own label.
What quality control measures are in place for Jodayu Cap?
Each batch undergoes rigorous testing, including HPLC, dissolution testing, and microbial analysis, to ensure compliance with pharmacopoeial standards.
How does your bulk supply process work?
Our bulk supply process includes customized packaging, logistics planning, and real-time tracking to ensure timely delivery of large orders.
Are there options for small-scale production?
Yes, we support small-scale production for trial runs or niche markets, with the same quality standards as bulk supply.
How can I inquire about bulk supply of Jodayu Cap?
For bulk supply inquiries, please contact us at **info@delwishealthcare.com** for tailored solutions and pricing.