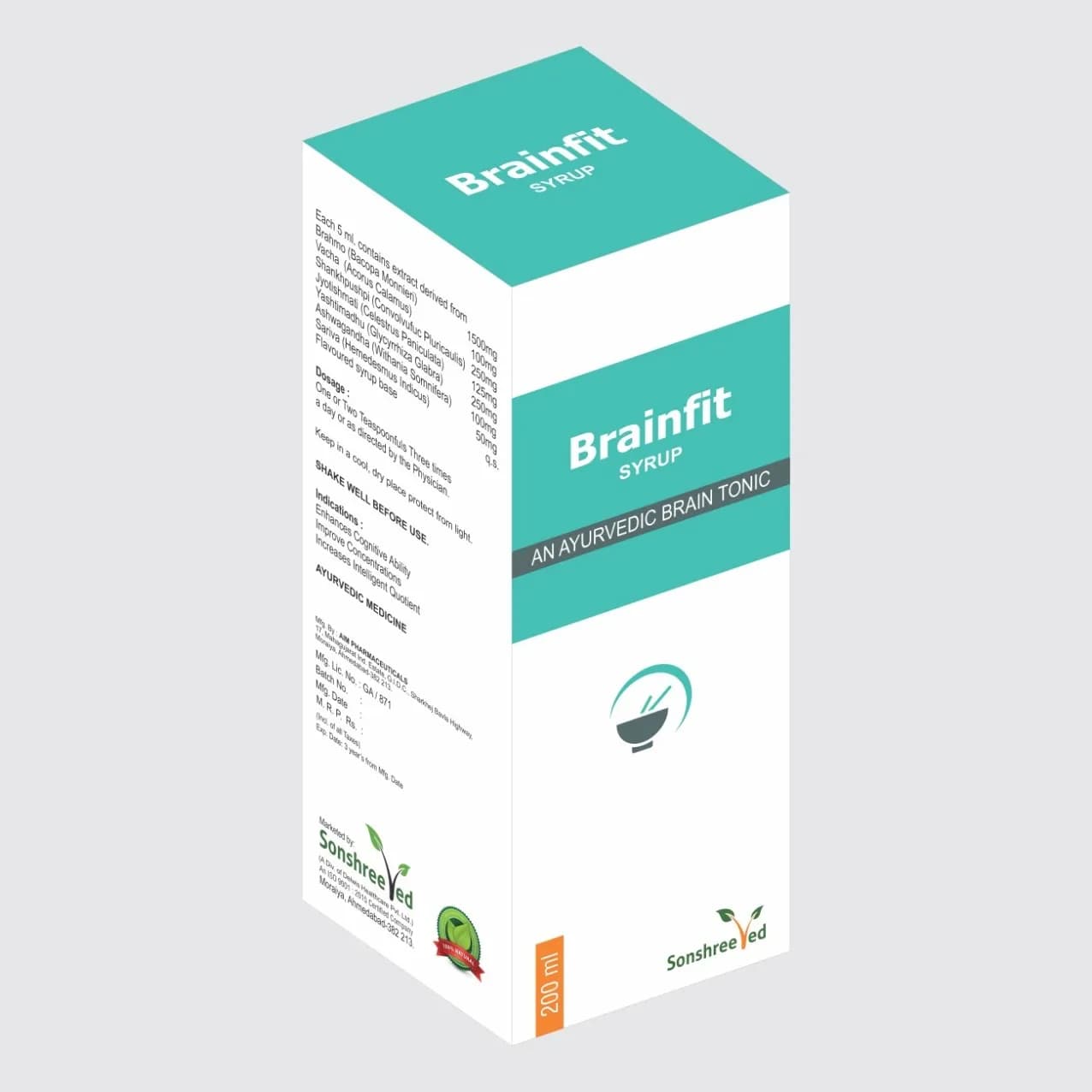
Liv It Syp is a proprietary formulation designed for pharmaceutical manufacturers, combining traditional herbal ingredients with modern excipients to ensure stability, solubility, and bioavailability. The core composition includes a standardized extract of Kalmegh (*Andrographis paniculata*), a well-documented herb known for its hepatoprotective properties, alongside a balanced blend of excipients to optimize shelf life and processing efficiency. The formulation is designed for oral administration, with a pH range of 5.5–6.5 to ensure compatibility with gastric fluids and minimize degradation of active ingredients. The product’s viscosity and flowability are tailored to meet tabletting and encapsulation requirements, while its stability profile is validated under accelerated storage conditions (40°C/75% RH) for 6 months. The excipient selection includes hydrophilic colloids, preservatives, and stabilizers to maintain product integrity during storage and transportation.
Manufacturing Capability
Our manufacturing infrastructure is equipped to produce Liv It Syp at scale, adhering to cGMP (current Good Manufacturing Practice) standards and ISO 9001:2015 quality management systems. The facility features a dedicated herbal extract processing unit, equipped with high-performance liquid chromatography (HPLC) systems for active ingredient quantification and a controlled environment for sterile processing. The production line supports batch sizes ranging from 500 to 5,000 liters, with scalability to accommodate bulk supply demands. All equipment is validated for process consistency, and the facility maintains a Class 10,000 cleanroom environment for critical steps such as filtration and filling. The manufacturing process includes rigorous in-process controls, including pH monitoring, microbial end-point testing, and particulate matter analysis, ensuring compliance with ICH Q7 and WHO guidelines for herbal medicines.
Available Strengths and Packaging
Composition: Liver Tonic enriched with Kalmegh.
Packaging: 200ml
Liv It Syp is available in a standardized 200ml formulation, designed for flexibility in dosage administration and packaging. The product’s concentration of Kalmegh extract is optimized for pharmaceutical applications, ensuring consistent potency across batches. The 200ml packaging is compatible with both glass and polyethylene terephthalate (PET) containers, with options for tamper-evident closures and child-resistant seals to meet regulatory requirements.
Contract Manufacturing / Third Party Manufacturing
As a trusted pharmaceutical manufacturer, we offer end-to-end contract manufacturing services for Liv It Syp, including private label production and third-party manufacturing for global distributors. Our facility is equipped to handle custom formulations, with the ability to adjust excipient ratios, active ingredient concentrations, and packaging specifications to meet specific client requirements. We support both small-scale pilot batches and large-volume commercial production, ensuring compliance with regulatory standards across markets. Our team of formulation scientists and quality assurance experts works closely with clients to optimize product specifications, from raw material sourcing to final packaging.
Quality Assurance
Quality assurance for Liv It Syp is governed by a comprehensive system of analytical validation, batch testing, and compliance with international pharmacopeia standards. Each batch undergoes rigorous testing for identity, purity, potency, and microbial contamination, using methods such as HPLC, UV spectrophotometry, and microbial limit testing. The product is validated against USP (United States Pharmacopeia) and EP (European Pharmacopoeia) monographs for herbal ingredients, ensuring consistency and safety. All raw materials are sourced from certified suppliers and tested for heavy metals, pesticides, and residual solvents. Finished product stability studies are conducted to confirm shelf life and storage conditions, with documentation traceable to ISO 17025 standards.
Regulatory Compliance
Liv It Syp complies with global regulatory frameworks, including cGMP, WHO guidelines for herbal medicines, and ISO 9001:2015 quality management systems. The product is manufactured in accordance with ICH Q7 guidelines for active pharmaceutical ingredients and meets the requirements of the FDA’s Current Good Manufacturing Practice (CGMP) for finished pharmaceuticals. Our facility is registered with the FDA, EMA, and CDSCO (Central Drug Standards Control Organization) for export to international markets. Compliance with ISO 22000:2018 for food safety and ISO 14001:2015 for environmental management ensures adherence to global sustainability standards. All documentation, including batch records and stability data, is maintained in accordance with regulatory audits and inspections.
Global Supply Capability
Our global supply chain is designed to meet the demands of pharmaceutical exporters and distributors, with the capacity to supply bulk quantities of Liv It Syp to over 50 countries. The facility is equipped for seamless export logistics, including compliance with IATA (International Air Transport Association) and IMDG (International Maritime Dangerous Goods) regulations for safe transportation. We maintain partnerships with international logistics providers to ensure timely delivery and adherence to customs requirements. Bulk supply capabilities range from 10,000 to 50,000 liters per month, with storage facilities compliant with WHO guidelines for temperature-sensitive products. Our supply chain reliability is supported by real-time tracking systems and a dedicated team for customs documentation and regulatory compliance.
Why Choose Us
Pharmaceutical distributors, exporters, and contract manufacturing partners select our facility for Liv It Syp due to our commitment to quality, scalability, and regulatory compliance. Our end-to-end manufacturing capabilities, combined with expertise in herbal formulation and global regulatory standards, ensure a reliable supply chain for international markets. The product’s standardized composition and compliance with ICH, USP, and WHO guidelines make it an ideal choice for bulk supply to healthcare providers and retailers. Our dedicated support team provides technical assistance, regulatory guidance, and customized solutions to meet the unique needs of each client.
Bulk Supply Inquiry
For bulk supply inquiries, contact our team at info@delwishealthcare.com to discuss customization, pricing, and logistics for Liv It Syp.
FAQs
What manufacturing standards does Liv It Syp adhere to?
Liv It Syp is manufactured in compliance with cGMP, ISO 9001:2015, and WHO guidelines for herbal medicines, ensuring quality and safety for global markets.
Can Liv It Syp be customized for different packaging formats?
Yes, our facility supports customization of packaging, including glass and PET containers, tamper-evident closures, and child-resistant seals, to meet regional regulatory requirements.
What is the shelf life of Liv It Syp under standard storage conditions?
Liv It Syp has a shelf life of 24 months when stored at 25°C ± 2°C and 60% ± 10% relative humidity.
Is Liv It Syp suitable for export to regulated markets like the EU and US?
Yes, the product is compliant with FDA CGMP, EMA guidelines, and ISO standards, making it suitable for export to the EU, US, and other regulated markets.
What quality control measures are in place for Liv It Syp?
Each batch undergoes HPLC, microbial testing, and stability studies to ensure potency, purity, and safety, as per ICH Q7 and USP standards.
Can Liv It Syp be produced in smaller batch sizes for pilot testing?
Yes, our facility supports small-scale production for pilot testing, with the ability to scale up to commercial volumes.
What certifications does your facility hold for pharmaceutical manufacturing?
Our facility is certified under cGMP, ISO 9001:2015, and ISO 14001:2015, with registration with FDA, EMA, and CDSCO.
How does your supply chain ensure timely delivery for international orders?
We partner with global logistics providers and maintain real-time tracking systems to ensure compliance with IATA and IMDG regulations for safe and timely delivery.
What is the minimum order quantity for bulk supply of Liv It Syp?
Bulk supply options start at 10,000 liters per month, with flexibility to adjust quantities based on client demand.
Can Liv It Syp be produced as a private label product?
Yes, we offer private label manufacturing services, allowing clients to brand the product under their own label while maintaining compliance and quality standards.