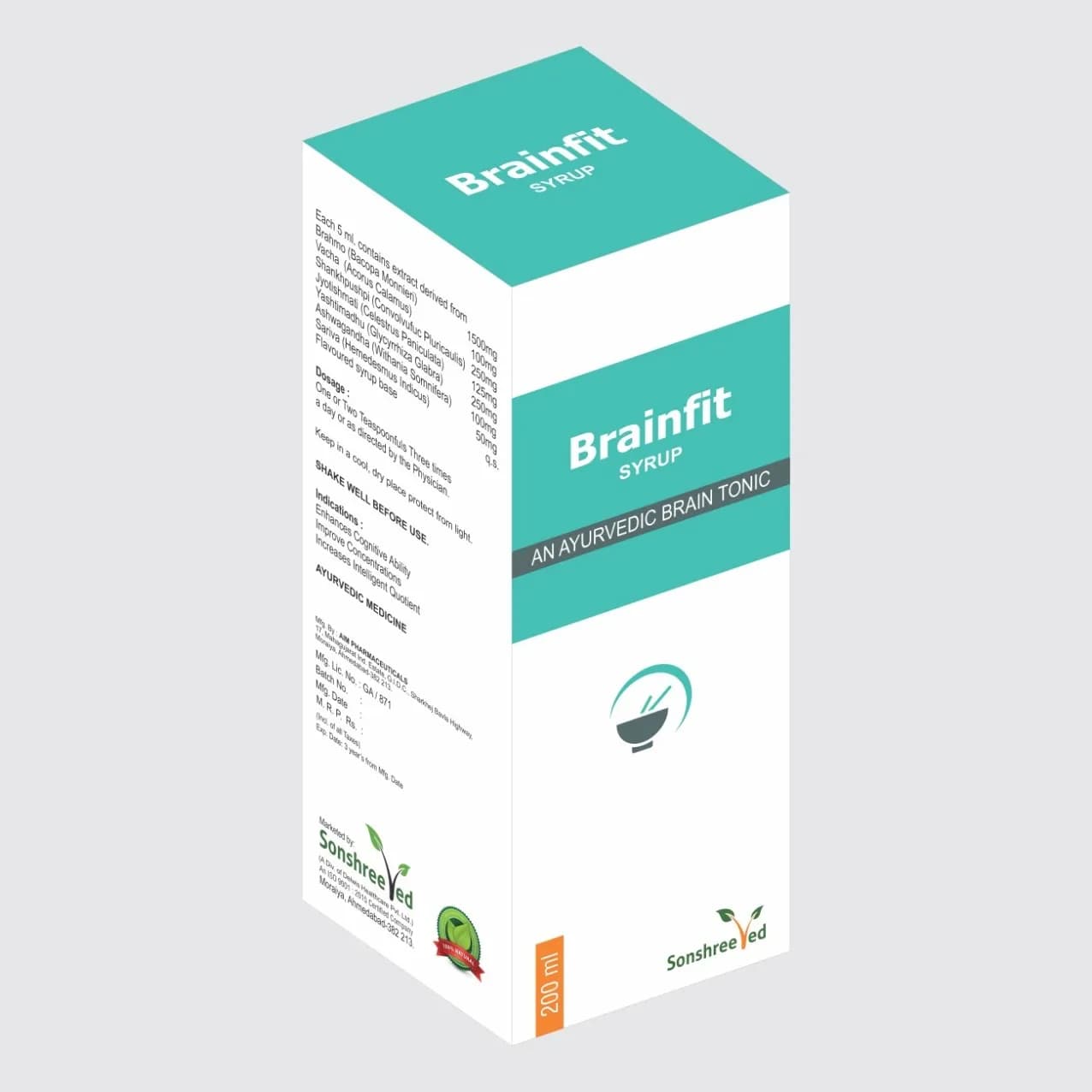
Lukone Syp is a specialized formulation engineered to address gynecological disorders, incorporating a proprietary blend of active ingredients and excipients tailored for stability, efficacy, and compatibility. The product’s formulation is designed to ensure consistent quality, with excipients selected for their ability to enhance solubility, prolong shelf life, and maintain the integrity of the active components under varied storage conditions. The manufacturing process focuses on optimizing the homogeneity of the liquid formulation, ensuring uniform dispersion of active ingredients for reliable performance. The product’s pharmaceutical characteristics include a pH-neutral base, which minimizes irritation and enhances patient compliance, while its viscosity is carefully controlled to facilitate accurate dosing. The formulation is designed to meet stringent pharmaceutical standards, with a focus on reproducibility and scalability to support bulk production requirements.
Manufacturing Capability
Delwis Healthcare’s manufacturing infrastructure is equipped to produce Lukone Syp with precision and efficiency, adhering to global pharmaceutical standards. The facility features advanced blending, homogenization, and filling systems to ensure consistent product quality. Sterile processing capabilities are integrated where necessary, with dedicated cleanrooms and aseptic equipment to prevent contamination. All manufacturing processes comply with Good Manufacturing Practice (GMP) guidelines, ensuring adherence to quality control protocols at every stage. The production line is designed for scalability, enabling seamless transitions from small-scale trials to large-volume commercial batches. Quality systems include real-time monitoring, automated data collection, and rigorous validation of equipment to maintain process reliability. The facility also employs robust documentation and traceability systems to support regulatory audits and ensure compliance with international standards.
Available Strengths and Packaging
Lukone Syp is formulated as a single strength, with the active ingredients and excipients calibrated to deliver optimal therapeutic outcomes. The product is available in a 200ml packaging format with an outer container, designed for safe transportation, storage, and bulk supply. The packaging ensures product integrity while meeting regulatory requirements for labeling, child-resistant features, and environmental sustainability.
Contract Manufacturing / Third Party Manufacturing
Delwis Healthcare offers comprehensive contract manufacturing and third-party manufacturing services tailored to the needs of pharmaceutical buyers. Our capabilities include private label manufacturing, where we produce Lukone Syp under a client’s brand name, ensuring full customization of packaging, labeling, and formulation. We also provide contract manufacturing for bulk supply, supporting both domestic and international markets. Our team collaborates closely with clients to align production schedules with demand forecasts, ensuring timely delivery without compromising quality. The facility is equipped to handle complex formulations and adhere to specific regulatory requirements, making it an ideal partner for pharmaceutical distributors and exporters seeking reliable, scalable production solutions.
Quality Assurance
Quality assurance at Delwis Healthcare is built on rigorous testing, analytical validation, and adherence to pharmaceutical standards. Every batch of Lukone Syp undergoes comprehensive testing for potency, purity, and stability, with results documented to ensure traceability. Analytical methods such as HPLC, UV spectrophotometry, and microbial testing are employed to validate the product’s consistency. The quality control system includes in-process checks, final product testing, and stability studies to confirm shelf life and performance under storage conditions. All documentation complies with ICH guidelines and regulatory expectations, ensuring transparency and reliability for buyers.
Regulatory Compliance
Delwis Healthcare ensures full compliance with Good Manufacturing Practice (GMP), World Health Organization (WHO) guidelines, and international ISO standards. Our facility is certified under ISO 9001:2015 for quality management and ISO 14001:2015 for environmental management, reflecting our commitment to sustainable and compliant operations. The manufacturing process adheres to WHO prequalification standards, enabling export to markets with stringent regulatory frameworks. Additionally, we maintain compliance with FDA, EMA, and other regional regulatory bodies, ensuring that Lukone Syp meets global quality and safety requirements.
Global Supply Capability
Delwis Healthcare is a trusted exporter with a robust international distribution network, capable of supplying Lukone Syp in bulk to markets across the globe. Our logistics partners ensure timely delivery through secure, temperature-controlled supply chains, supporting both short-term and long-term contracts. The facility is equipped to handle large-scale production, with storage capabilities and packaging lines designed for high-volume orders. Supply chain reliability is maintained through real-time tracking systems, inventory management, and contingency planning to mitigate disruptions. This ensures consistent availability of Lukone Syp for pharmaceutical distributors and exporters, regardless of market demand fluctuations.
Why Choose Us
Pharmaceutical distributors, exporters, and contract manufacturing partners benefit from Delwis Healthcare’s expertise in producing high-quality, compliant products like Lukone Syp. Our commitment to GMP, ISO certification, and regulatory compliance ensures that buyers meet global standards without compromising on quality. The facility’s scalability and advanced manufacturing capabilities enable efficient bulk supply, while our dedicated support team provides end-to-end assistance from production to delivery. Choosing Delwis Healthcare means partnering with a reliable manufacturer that prioritizes innovation, sustainability, and customer satisfaction.
Bulk Supply Inquiry
Delwis Healthcare offers seamless bulk supply solutions for Lukone Syp. For inquiries regarding large-scale procurement, production timelines, or customized packaging, contact our team at info@delwishealthcare.com.
FAQs
What is the manufacturing process for Lukone Syp?
Lukone Syp is produced using advanced blending, homogenization, and filling systems, ensuring uniformity and compliance with GMP standards.
Can Lukone Syp be customized for private label manufacturing?
Yes, Delwis Healthcare offers private label manufacturing services, allowing clients to brand Lukone Syp under their own label.
What certifications does Delwis Healthcare hold?
Our facility is certified under ISO 9001:2015, ISO 14001:2015, and adheres to WHO and FDA guidelines for global compliance.
Is Lukone Syp suitable for bulk supply?
Yes, the product is designed for scalable production, with packaging and logistics optimized for bulk distribution.
What quality control measures are in place for Lukone Syp?
Every batch undergoes rigorous testing, including HPLC, microbial analysis, and stability studies, to ensure consistency and safety.
Can Lukone Syp be exported to international markets?
Yes, our compliance with WHO, FDA, and EMA standards enables seamless export to global markets.
How does Delwis Healthcare ensure supply chain reliability?
We employ real-time tracking, inventory management, and contingency planning to guarantee consistent supply.
What is the shelf life of Lukone Syp?
The product’s shelf life is validated through stability studies, ensuring optimal performance within specified storage conditions.
Can Delwis Healthcare accommodate urgent bulk orders?
Yes, our scalable production capabilities allow for expedited manufacturing to meet urgent supply needs.
How can I request a sample or bulk supply of Lukone Syp?
Contact our team at **info@delwishealthcare.com** for assistance with samples, bulk orders, or customized solutions.