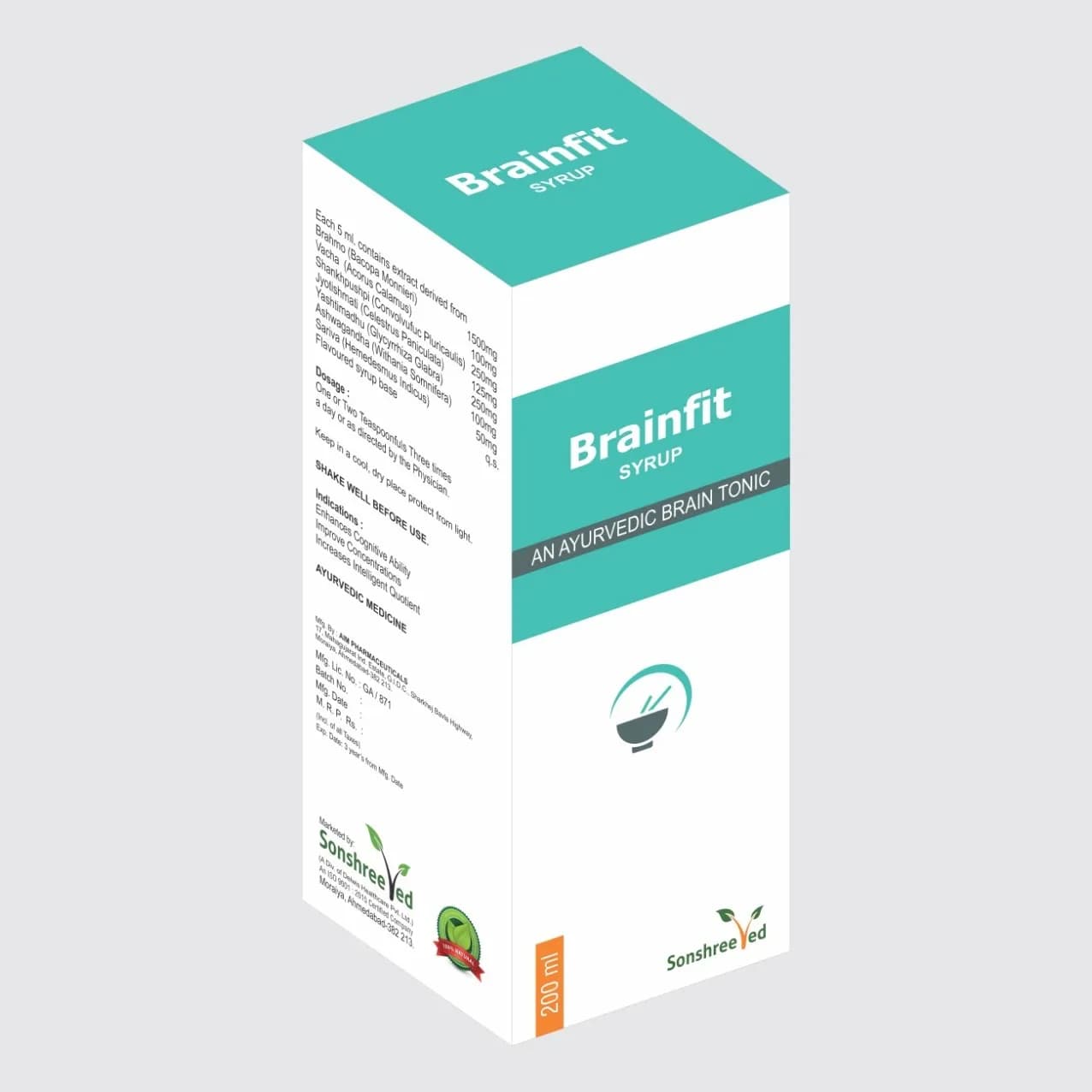
Uril Syrup is a meticulously formulated pharmaceutical tonic designed for Renal and Urinary Tract infections, combining standardized herbal extracts with excipients to ensure stability, bioavailability, and compliance with pharmaceutical specifications. The formulation employs a proprietary blend of active botanicals, carefully selected for their established roles in urinary tract health, alongside excipients that enhance solubility, preservation, and shelf life. The manufacturing process involves precise dissolution, homogenization, and filtration steps to ensure uniformity in active ingredient concentration and physical consistency. The syrup’s viscous texture and sweetening agents are optimized for ease of administration while maintaining microbial stability. Key pharmaceutical characteristics include a pH-neutral formulation, preservative compatibility, and adherence to ICH guidelines for excipient selection. The product’s formulation is designed to meet the demands of bulk manufacturing, ensuring scalability without compromising quality or potency.
Manufacturing Capability
Delwis Healthcare’s manufacturing infrastructure is engineered to deliver high-quality pharmaceutical products like Uril Syrup, supported by state-of-the-art facilities compliant with current Good Manufacturing Practices (cGMP). The facility is equipped with advanced sterile processing capabilities, including aseptic filling lines for non-sterile products, ensuring contamination-free production. Our production lines are designed for scalability, enabling seamless transition from small-scale trials to large-volume commercial batches. The manufacturing process adheres to strict quality control protocols, including real-time monitoring of temperature, humidity, and particle count during formulation and packaging. Our quality systems integrate Hazard Analysis and Critical Control Points (HACCP) principles, ensuring compliance with global pharmaceutical standards. Additionally, the facility is certified for ISO 9001:2015 quality management and maintains rigorous documentation for traceability, batch validation, and regulatory audits.
Available Strengths and Packaging
Uril Syrup is formulated as a standardized tonic for Renal and Urinary Tract infections, available in a single strength optimized for therapeutic efficacy. The product is packaged in 200ml bottles with an outer carton, ensuring secure transportation and tamper-evidence. The packaging design prioritizes durability, compliance with international shipping regulations, and consumer safety. The 200ml volume is tailored for bulk supply and retail distribution, balancing portability with adequate dosage per container.
Contract Manufacturing / Third Party Manufacturing
As a trusted pharmaceutical manufacturer, Delwis Healthcare offers comprehensive contract manufacturing and third-party production services tailored to the needs of global buyers. Our private label manufacturing capabilities enable clients to produce branded products under their own trademarks, with full flexibility in formulation, packaging, and labeling. We support a wide range of pharmaceutical requirements, including API sourcing, excipient integration, and custom dosage forms. Our team of expert formulation scientists and regulatory specialists ensures compliance with client-specific specifications and international standards. Whether you require small-batch prototyping or large-scale commercial production, our scalable infrastructure and dedicated project management team deliver timely, cost-effective solutions for pharmaceutical buyers.
Quality Assurance
Delwis Healthcare’s quality assurance framework is built on rigorous analytical validation, batch testing, and adherence to pharmaceutical quality standards. Every production batch undergoes comprehensive testing for pH, viscosity, microbial limits, and active ingredient potency, ensuring compliance with USP, EP, and BP monographs. Advanced analytical techniques, including HPLC and spectrophotometry, validate the consistency of herbal extracts and excipient compatibility. Our quality control systems are integrated with real-time data monitoring, enabling immediate corrective actions for deviations. All finished products are subjected to stability testing under accelerated conditions to guarantee shelf life and efficacy. Additionally, our facility maintains a robust traceability system, allowing full documentation of raw material sourcing, manufacturing steps, and distribution channels for regulatory audits.
Regulatory Compliance
Delwis Healthcare ensures full compliance with global regulatory frameworks, including cGMP, WHO guidelines, and ISO 9001:2015 standards. Our manufacturing processes adhere to the latest ICH guidelines for quality risk management and analytical method validation. The facility is registered with regulatory authorities in key markets, including the US FDA, EU MDR, and ASEAN regulatory bodies, enabling seamless export to over 150 countries. Our quality management system is regularly audited by third-party agencies to maintain compliance with international benchmarks. Additionally, we support regulatory submissions for market approval, including dossier preparation, labeling compliance, and Good Distribution Practice (GDP) adherence.
Global Supply Capability
Delwis Healthcare specializes in bulk supply and global distribution of pharmaceutical products, including Uril Syrup, with a robust international logistics network. Our export capabilities extend to major pharmaceutical markets in Asia, Africa, Latin America, and the Middle East, supported by partnerships with trusted logistics providers and customs clearance experts. The 200ml packaging is optimized for bulk supply, ensuring cost-effective transportation and storage. Our supply chain infrastructure prioritizes reliability, with redundant inventory management systems and real-time tracking to mitigate disruptions. We offer flexible supply terms, including long-term contracts and just-in-time delivery, to meet the dynamic needs of pharmaceutical buyers.
Why Choose Us
Pharmaceutical distributors, exporters, and contract manufacturing partners select Delwis Healthcare for our unmatched expertise in quality manufacturing, regulatory compliance, and global supply chain efficiency. Our commitment to innovation and adherence to international standards ensures that products like Uril Syrup meet the highest benchmarks for safety and efficacy. With a proven track record of delivering bulk supplies to over 50 countries, we provide end-to-end support, from formulation to market entry. Our dedicated team of pharmacists, scientists, and logistics experts ensures seamless collaboration, enabling clients to focus on growth while we manage the complexities of production and distribution.
Bulk Supply Inquiry
Delwis Healthcare offers reliable bulk supply of Uril Syrup, tailored to meet the demands of pharmaceutical buyers. For inquiries regarding large-scale procurement, please contact us at info@delwishealthcare.com.
FAQs
What certifications does Delwis Healthcare hold for pharmaceutical manufacturing?
Our facility is certified under cGMP, ISO 9001:2015, and WHO GMP, ensuring compliance with global regulatory standards.
Can Uril Syrup be customized for private label manufacturing?
Yes, we offer private label manufacturing services, allowing clients to brand the product under their own trademark.
What are the packaging options for Uril Syrup?
The product is available in 200ml bottles with an outer carton, designed for secure transportation and retail display.
Does Delwis Healthcare support export to international markets?
Yes, we have a global distribution network and regulatory registrations in over 150 countries, enabling seamless international exports.
How does Delwis ensure quality consistency in bulk supply?
Every batch undergoes rigorous testing for potency, microbial limits, and stability, with full traceability for regulatory compliance.
Can Uril Syrup be produced in different strengths?
While the product is standardized for Renal and Urinary Tract infections, we can adjust formulations based on client specifications.
What is the minimum order quantity for bulk supply?
We accommodate flexible order quantities, with minimums tailored to the scale of your pharmaceutical requirements.
How does Delwis handle regulatory submissions for market approval?
Our team assists with dossier preparation, labeling compliance, and regulatory submissions to expedite market entry.
What is the shelf life of Uril Syrup?
The product has a shelf life of 24 months when stored under recommended conditions, with stability testing confirming efficacy.
Can Delwis Healthcare provide samples for quality evaluation?
Yes, we offer samples for evaluation, with full documentation of formulation details and compliance certifications.