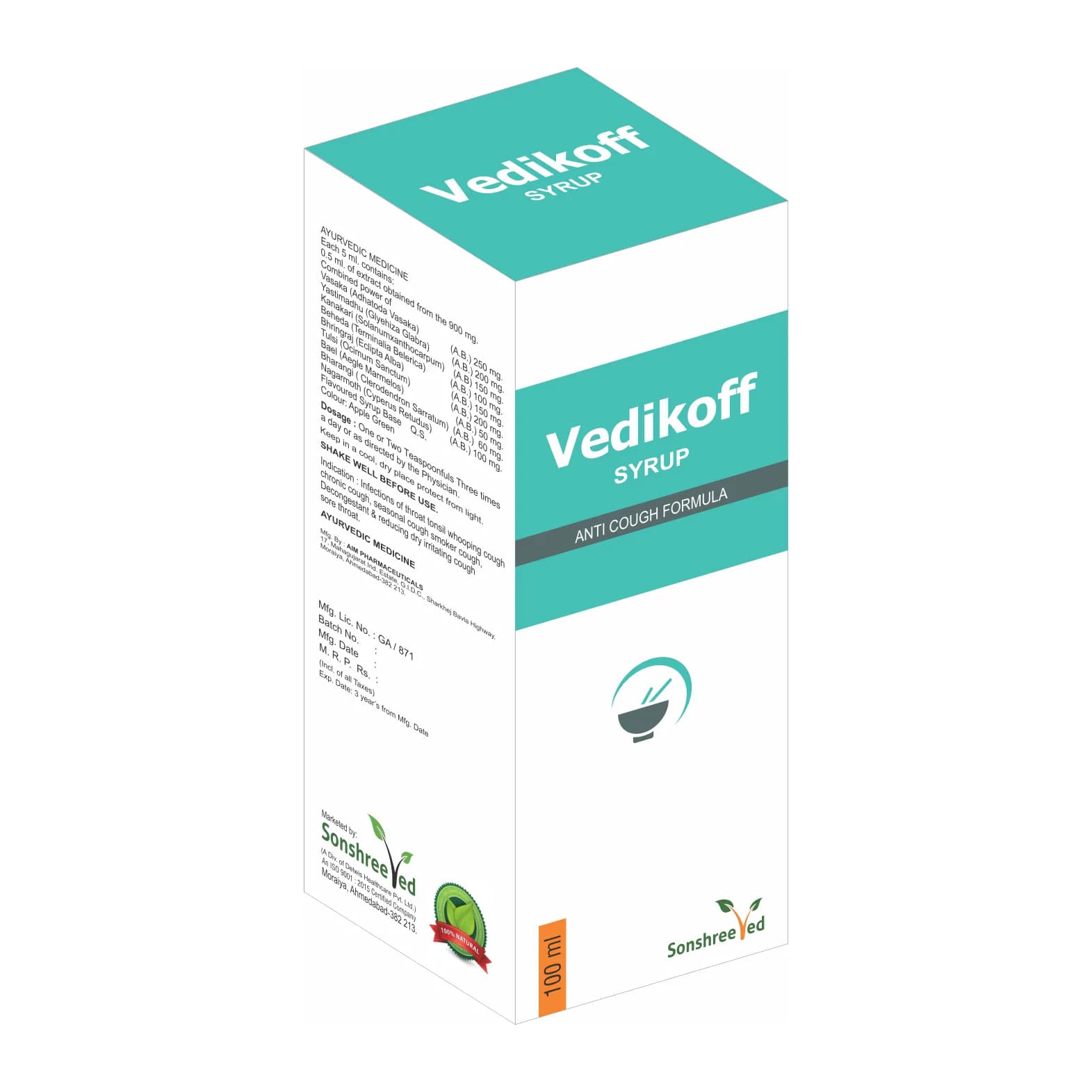
Vedikoff Syp is a meticulously formulated Ayurvedic cough syrup designed for pharmaceutical applications, incorporating a blend of traditional herbal ingredients with Tulsi (Ocimum sanctum) as a key active component. The formulation leverages Ayurvedic principles to create a stable, homogeneous liquid dosage form suitable for commercial production. The syrup’s base includes purified excipients, preservatives, and flavoring agents to ensure consistency, shelf stability, and ease of administration. The manufacturing process involves precise blending, homogenization, and sterilization techniques to maintain product integrity. The inclusion of Tulsi, renowned for its expectorant and anti-inflammatory properties, is standardized to ensure therapeutic efficacy. The formulation adheres to pharmaceutical-grade specifications, ensuring compatibility with modern dosage form requirements while retaining the therapeutic synergy of Ayurvedic ingredients.
Manufacturing Capability
Our manufacturing infrastructure is designed to meet the rigorous demands of pharmaceutical production, with a focus on scalability, sterility, and compliance. The facility is equipped with state-of-the-art equipment for liquid formulation, including high-speed blending systems, homogenizers, and aseptic filling lines to ensure contamination-free processing. Sterile processing capabilities are maintained through ISO-certified cleanrooms and validated sterilization protocols, ensuring adherence to GMP standards. Production scalability is achieved through modular manufacturing units that can accommodate varying batch sizes, from small-scale trials to large-volume commercial runs. The facility’s quality systems include real-time monitoring, automated data logging, and rigorous validation of critical process parameters. All manufacturing activities are conducted under strict GMP guidelines, ensuring consistency, traceability, and compliance with international regulatory frameworks.
Available Strengths and Packaging
Composition: Ayurvedic Cough Syrup enriched with Tulsi.
Packaging: 100ml With Outer
Vedikoff Syp is formulated with a standardized strength to ensure consistent therapeutic efficacy. The syrup’s concentration of Tulsi and other Ayurvedic ingredients is optimized for stability and potency. The product is packaged in 100ml bottles with outer packaging, designed for secure transportation and storage. The outer packaging meets pharmaceutical packaging standards, providing protection against environmental factors while ensuring compliance with labeling and regulatory requirements.
Contract Manufacturing / Third Party Manufacturing
As a trusted manufacturer, we offer comprehensive contract manufacturing and third-party production services tailored to pharmaceutical buyers’ needs. Our capabilities include private label manufacturing, where we produce finished products under a client’s brand name, ensuring full customization of packaging, labeling, and formulation specifications. We also provide flexible contract manufacturing options, enabling clients to leverage our expertise in Ayurvedic formulation while maintaining control over their supply chain. Our services are designed to meet global regulatory standards, with the ability to scale production to match demand. Whether you require a single batch or large-scale commercial production, our team ensures seamless execution, compliance, and quality assurance throughout the manufacturing lifecycle.
Quality Assurance
Our quality assurance framework is built on rigorous testing, analytical validation, and adherence to pharmaceutical standards. Every batch of Vedikoff Syp undergoes comprehensive quality control checks, including pH testing, microbial limit testing, and stability studies to ensure product integrity. Analytical validation is conducted using HPLC (High-Performance Liquid Chromatography) and other advanced techniques to confirm the consistency of active ingredients, including Tulsi. Batch testing is performed at multiple stages of production to identify and mitigate deviations. Our quality systems are aligned with GMP, WHO, and ISO standards, ensuring that all products meet international benchmarks for safety, efficacy, and purity.
Regulatory Compliance
Vedikoff Syp is manufactured in full compliance with global regulatory frameworks, including GMP, WHO guidelines, and ISO standards. Our facility is certified under ISO 9001:2015 for quality management and ISO 22000 for food safety, ensuring adherence to stringent operational protocols. Compliance with WHO guidelines ensures that the product meets international standards for herbal pharmaceuticals, while GMP certification guarantees adherence to manufacturing best practices. We also maintain documentation for regulatory submissions, including dossiers for export markets, ensuring seamless approval processes in target regions. Our commitment to regulatory compliance extends to environmental and labor standards, reflecting our dedication to sustainable and ethical pharmaceutical practices.
Global Supply Capability
We offer robust global supply capabilities, including export-ready manufacturing, international distribution networks, and bulk supply solutions. Our logistics partners ensure reliable delivery to over 50 countries, with customized shipping options to meet regional requirements. Bulk supply capacity is supported by scalable production lines and inventory management systems, enabling timely fulfillment of large orders. The supply chain is designed for resilience, with redundant systems to mitigate disruptions and ensure continuity. Our export compliance team ensures adherence to customs regulations, import documentation, and phytosanitary requirements, facilitating smooth international trade. Whether you require small batches or large-scale distribution, our global supply chain infrastructure ensures timely and cost-effective delivery.
Why Choose Us
Pharmaceutical distributors, exporters, and contract manufacturing partners select us for our unparalleled expertise in Ayurvedic pharmaceuticals, compliance-driven manufacturing, and global supply capabilities. Our commitment to GMP, ISO, and WHO standards ensures that Vedikoff Syp meets the highest quality benchmarks. The ability to scale production, coupled with flexible contract manufacturing options, positions us as a reliable partner for diverse market needs. Our global distribution network and export-ready infrastructure enable seamless international trade, while our focus on sustainability and ethical practices aligns with modern industry expectations. By partnering with us, you gain access to a trusted manufacturer with the capacity to deliver high-quality, compliant products to global markets.
Bulk Supply Inquiry
We provide bulk supply capabilities for Vedikoff Syp, tailored to meet the demands of pharmaceutical buyers. For inquiries regarding large-scale procurement, please contact:
info@delwishealthcare.com
FAQs
What manufacturing standards does Vedikoff Syp comply with?
Vedikoff Syp is manufactured in compliance with GMP, WHO guidelines, and ISO standards, ensuring adherence to international pharmaceutical quality benchmarks.
Can Vedikoff Syp be customized for private label manufacturing?
Yes, we offer private label manufacturing services, allowing clients to brand the product under their own name while maintaining quality and compliance.
What is the shelf life of Vedikoff Syp?
The product has a shelf life of 24 months when stored under recommended conditions, as validated through stability testing.
Is Vedikoff Syp suitable for export to international markets?
Yes, our export-ready manufacturing and compliance framework ensure seamless regulatory approval and distribution in global markets.
What packaging options are available for Vedikoff Syp?
The product is available in 100ml bottles with outer packaging, designed for secure transportation and compliance with labeling regulations.
Can Vedikoff Syp be produced in larger quantities?
Yes, our scalable manufacturing infrastructure supports bulk production to meet varying demand levels.
What quality control measures are in place for Vedikoff Syp?
Each batch undergoes rigorous testing, including microbial limit tests, pH analysis, and HPLC validation to ensure consistency and purity.
How does your supply chain ensure reliability?
Our logistics network and inventory management systems ensure timely delivery, with redundant systems to mitigate supply chain disruptions.
Are there options for contract manufacturing beyond Vedikoff Syp?
Yes, we offer contract manufacturing services for a range of Ayurvedic and pharmaceutical products, tailored to client specifications.
How can I request a bulk supply of Vedikoff Syp?
For bulk supply inquiries, please contact us at **info@delwishealthcare.com** to discuss your requirements and procurement details.