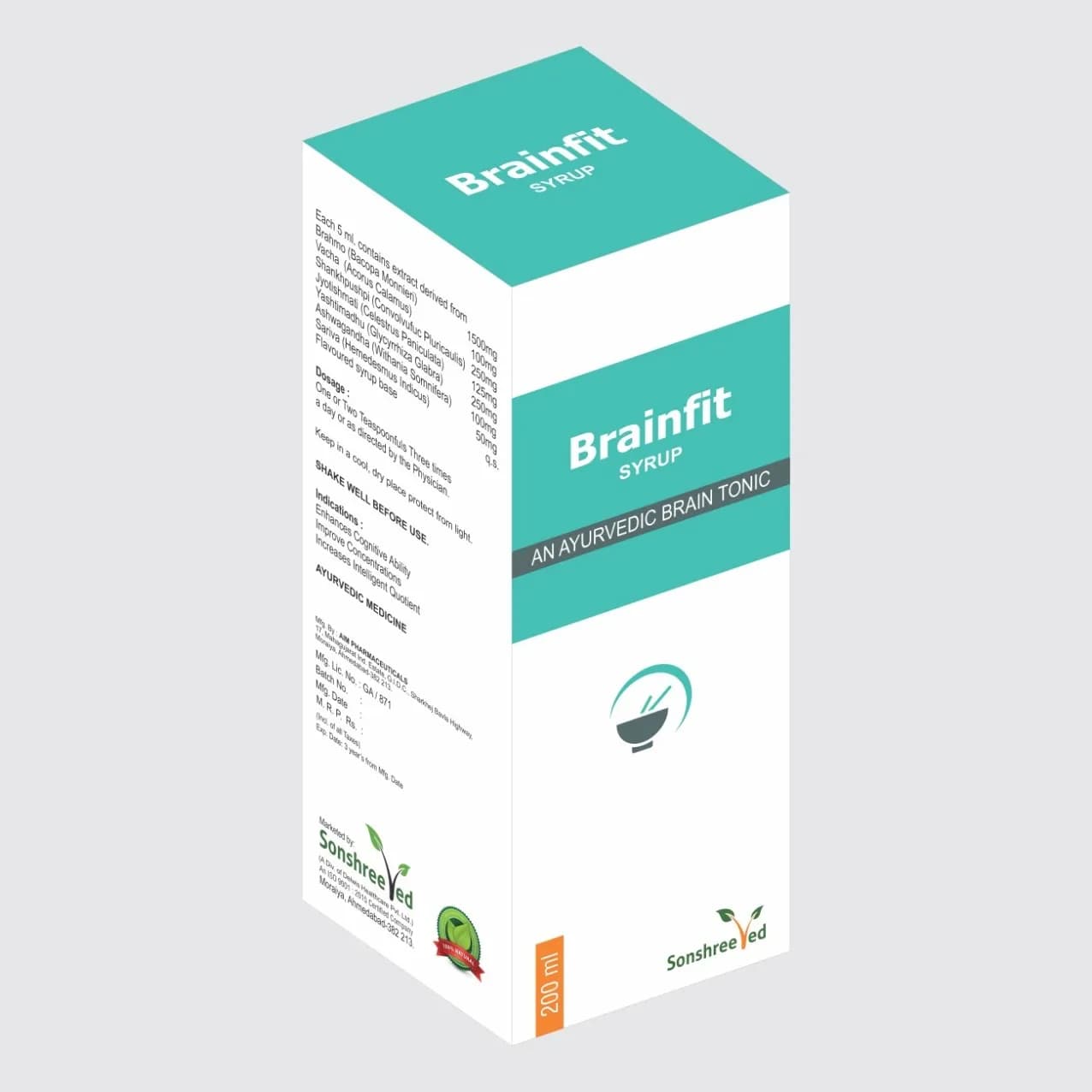
Bonjo - CQ Cap is a meticulously formulated herbal capsule designed for pharmaceutical manufacturers requiring advanced therapeutic solutions. The formulation integrates three key active ingredients: Hadjod (Cissus Quadrangularis) at 400mg, Kukudwatak Bhashma at 60mg, and Ashwagandha (Withania Somnifera) at 35mg, each selected for its pharmacological properties and synergistic potential. Hadjod is renowned for its role in bone regeneration and tissue repair, while Kukudwatak Bhashma contributes to mineralization and metabolic support. Ashwagandha enhances cellular resilience and anti-inflammatory effects, collectively supporting the formulation’s therapeutic profile. Sodium Benzoate IP at 5mg ensures microbial stability, maintaining product integrity during storage and transportation. The capsule’s composition is optimized for uniformity, dissolution rate, and bioavailability, aligning with pharmaceutical standards for active pharmaceutical ingredients (APIs) and excipients. The formulation’s stability under varying environmental conditions ensures consistent quality, making it suitable for large-scale production and global distribution.
Manufacturing Capability
Our manufacturing infrastructure is designed to meet the stringent demands of pharmaceutical production, with state-of-the-art facilities equipped for sterile processing, bulk API handling, and high-throughput capsule manufacturing. The facility adheres to Good Manufacturing Practice (GMP) standards, ensuring compliance with international regulatory frameworks. Advanced machinery, including high-speed capsule filling lines and automated tablet compression systems, enables precise dosing and consistent product quality. The production process incorporates quality control checkpoints at every stage, from raw material sourcing to final packaging, ensuring adherence to specifications. Scalability is a core strength, with the capability to produce large batches while maintaining uniformity in composition and physical attributes. The facility’s quality systems include validated cleaning procedures, environmental monitoring, and real-time data analytics to mitigate contamination risks. Additionally, the infrastructure supports customization for private-label manufacturing, allowing pharmaceutical buyers to integrate the product into their portfolio with minimal logistical overhead.
Available Strengths and Packaging
Bonjo - CQ Cap is available in a single standardized strength, with the composition fixed at 400mg Hadjod, 60mg Kukudwatak Bhashma, 35mg Ashwagandha, and 5mg Sodium Benzoate IP. The product is packaged in 6x10 Alu Alu blister packs, ensuring tamper-evident sealing, moisture resistance, and compliance with pharmaceutical packaging regulations. The Alu Alu material provides optimal protection against light, heat, and humidity, preserving the integrity of the active ingredients during transit and storage. This packaging format is ideal for bulk supply and retail distribution, offering convenience for end-users while meeting global pharmaceutical packaging standards.
Contract Manufacturing / Third Party Manufacturing
As a trusted manufacturer, we offer comprehensive contract manufacturing and third-party production services tailored to pharmaceutical buyers’ needs. Our capabilities include private-label manufacturing, where we produce finished products under a client’s brand, ensuring full customization of packaging, labeling, and quality control protocols. For contract manufacturing, we provide end-to-end solutions, from API sourcing to final packaging, with flexibility to accommodate varying specifications and production scales. Our facility is equipped to handle both small and large-volume orders, supporting clients in meeting market demands without compromising quality. Additionally, we offer outsourcing services for API synthesis, formulation development, and stability testing, enabling pharmaceutical companies to leverage our expertise while maintaining control over their product lifecycle.
Quality Assurance
Our quality assurance framework is built on rigorous analytical validation, batch testing, and compliance with global pharmaceutical standards. Every production batch undergoes extensive testing, including high-performance liquid chromatography (HPLC) for API quantification, microbial limit testing, and dissolution profiling to ensure bioavailability. Raw materials are sourced from certified suppliers and subjected to in-process testing for purity and potency. The facility’s quality control systems are validated to meet ICH and USP guidelines, ensuring consistency in product performance. Documentation, including batch records and stability data, is maintained for traceability and regulatory audits. Continuous improvement initiatives, such as Six Sigma and Lean manufacturing, further enhance process efficiency and product reliability.
Regulatory Compliance
We adhere to stringent regulatory frameworks, including Good Manufacturing Practice (GMP), World Health Organization (WHO) guidelines, and International Organization for Standardization (ISO) standards. Our facility is certified under ISO 9001 for quality management and ISO 14001 for environmental management, reflecting our commitment to sustainability and operational excellence. Compliance with GMP ensures that all manufacturing processes meet international safety and efficacy benchmarks. Additionally, we maintain compliance with the United States Pharmacopeia (USP) and European Pharmacopeia (EP) standards for API purity and product specifications. Our regulatory expertise extends to FDA, EMA, and WHO guidelines, enabling seamless export to global markets. Regular audits and documentation ensure alignment with evolving regulatory requirements, providing pharmaceutical buyers with confidence in product legitimacy.
Global Supply Capability
Our global supply chain is designed to support bulk exports, international distribution, and reliable bulk supply to pharmaceutical markets worldwide. With a well-established network of logistics partners, we ensure timely delivery to destinations across Asia, Africa, Europe, and the Americas. The facility’s capacity for large-scale production allows for consistent bulk supply, meeting the demands of both regional and international buyers. Our inventory management systems and forecasting tools optimize supply chain efficiency, minimizing delays and ensuring product availability. Additionally, our compliance with international shipping regulations and phytosanitary standards facilitates seamless cross-border transactions. The ability to scale production in response to market fluctuations further strengthens our position as a dependable supplier for global pharmaceutical distributors.
Why Choose Us
Pharmaceutical distributors, exporters, and contract manufacturing partners select us for our expertise in herbal formulation, compliance-driven manufacturing, and scalable production capabilities. Our facility’s adherence to GMP, ISO, and WHO standards ensures product quality and regulatory readiness for global markets. The ability to customize packaging, branding, and production specifications allows clients to meet diverse market demands without compromising on quality. Our commitment to innovation, coupled with a proven track record in bulk supply, positions us as a reliable partner for businesses seeking to expand their product portfolios. By leveraging our infrastructure and regulatory expertise, clients can achieve cost-efficiency, compliance assurance, and market competitiveness.
Bulk Supply Inquiry
For bulk supply inquiries, please contact us at info@delwishealthcare.com. Our team is dedicated to providing tailored solutions for pharmaceutical manufacturers, exporters, and distributors seeking high-quality, compliant herbal formulations.
FAQs
What certifications does your facility hold?
Our facility is certified under ISO 9001, ISO 14001, and adheres to GMP, WHO, and FDA guidelines.
Can you customize packaging formats?
Yes, we offer customization for packaging, including 6x10 Alu Alu blister packs and other formats upon request.
What is the minimum order quantity for bulk supply?
We accommodate both small and large orders, with bulk supply capabilities for global distribution.
Do you provide API sourcing services?
Yes, we source high-purity APIs and offer formulation development support for private-label manufacturing.
How do you ensure product stability?
Our formulation includes Sodium Benzoate IP as a preservative, and packaging is designed for moisture and light resistance.
Can you support export to specific regions?
Yes, we comply with international shipping regulations and have a global distribution network for seamless exports.
What quality control measures are in place?
Each batch undergoes HPLC, microbial testing, and dissolution profiling to ensure compliance with USP and EP standards.
How do you handle regulatory audits?
We maintain comprehensive documentation and conduct internal audits to ensure compliance with FDA, EMA, and WHO guidelines.
Can you produce under private-label branding?
Yes, we offer private-label manufacturing with full customization of packaging, labeling, and quality control protocols.
What is your lead time for bulk orders?
Lead times vary based on order size, but we prioritize timely delivery to meet client deadlines and market demands.