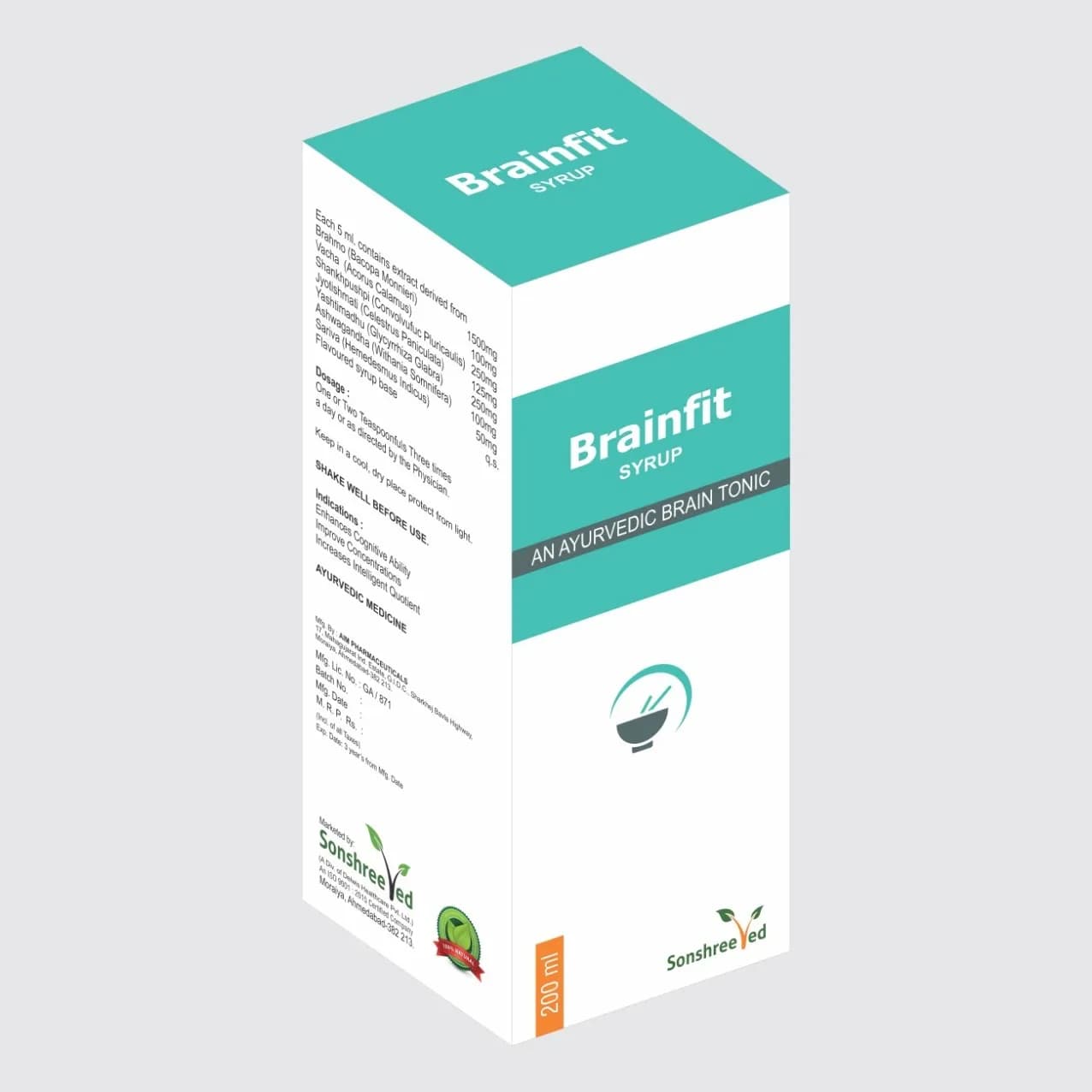
Piloherb Cap is a hard gelatin capsule formulation designed for pharmaceutical applications, containing a precise blend of standardized herbal and Ayurvedic ingredients. The composition includes Harde (Terminalia chebula) at 50mg, Suddha Sonageru (R.T.S.-1) at 45mg, Rasvanti (Berberis aristata) at 45mg, Kumari (Aloe indica) at 45mg, Aritha (Sapnitus trifoliatus) at 45mg, Neem (Azadirachta indica) at 45mg, Kala Mari (Piper nigrum) at 45mg, Suvarna Makshik Bhasma (R.T.S.-1) at 45mg, Galo (Tinospora cordifolia) at 45mg, and Garmalo (Cassia fistula) at 45mg. The formulation is designed to ensure stability, uniformity, and compatibility of active ingredients, with each component selected for its pharmacological properties and synergistic potential. The hard gelatin capsule form provides controlled release and protection of sensitive phytoconstituents, while maintaining bioavailability. Manufacturing adheres to strict excipient compatibility protocols to ensure no adverse interactions, with all components processed to meet pharmaceutical-grade specifications.
Manufacturing Capability
Our pharmaceutical manufacturing infrastructure is equipped with state-of-the-art facilities compliant with cGMP, ISO 9001, and WHO guidelines. The facility includes advanced capsule filling lines, drying chambers, and packaging systems capable of handling high-volume production. Sterile processing capabilities are maintained through HEPA-filtered environments and validated cleaning protocols. Production scalability is ensured through modular manufacturing units, allowing for flexible batch sizes to meet global demand. Quality systems are integrated throughout the manufacturing process, with real-time monitoring of critical parameters such as temperature, humidity, and particle size. The facility is designed to support both small-scale prototyping and large-scale bulk production, ensuring consistency in formulation and adherence to regulatory standards.
Available Strengths and Packaging
Piloherb Cap is formulated with a standardized strength of 50mg for Harde (Terminalia chebula) and 45mg for all other active ingredients, ensuring consistent potency across batches. The product is packaged in 10x10 Alu Alu blister packs, providing tamper-evident, moisture-resistant, and light-protected storage. This packaging format is ideal for both retail and bulk distribution, with the Alu Alu material offering durability and compliance with pharmaceutical packaging regulations.
Contract Manufacturing / Third Party Manufacturing
As a trusted manufacturer, we offer comprehensive contract manufacturing, private label, and third-party manufacturing services tailored to pharmaceutical buyers. Our capabilities include formulation development, API integration, and full-scale production of customized dosage forms. We support clients in meeting regional and international regulatory requirements, with flexibility to adapt formulations to specific market needs. Our team provides end-to-end solutions, from raw material sourcing to finished product packaging, ensuring compliance with cGMP, ISO, and WHO standards.
Quality Assurance
Our quality assurance framework is built on rigorous analytical validation, batch testing, and compliance with global pharmacopeia standards. Each batch undergoes testing for identity, purity, potency, and dissolution using advanced techniques such as HPLC, UV spectrophotometry, and microbial limit testing. Quality control measures include in-process checks, stability testing, and third-party audits to ensure consistency and safety. All raw materials are sourced from certified suppliers and tested for heavy metals, pesticides, and microbial contaminants. Our systems ensure traceability, documentation, and adherence to pharmacopeial monographs, guaranteeing product reliability for pharmaceutical buyers.
Regulatory Compliance
We adhere to stringent regulatory frameworks, including cGMP, WHO Good Manufacturing Practices (GMP), and ISO 9001 certification. Our manufacturing processes comply with global standards for pharmaceutical production, ensuring products meet the requirements of regulatory authorities such as the FDA, EMA, and PMDA. Documentation is maintained in accordance with ICH guidelines, and all products are registered with relevant regulatory bodies. Compliance with ISO 14001 environmental standards further ensures sustainable and responsible manufacturing practices.
Global Supply Capability
Our global supply capability is supported by an extensive international distribution network, enabling seamless export to over 50 countries. We offer bulk supply options tailored to meet the demands of pharmaceutical distributors, exporters, and contract manufacturers. Our logistics partners ensure timely delivery, with cold chain solutions for temperature-sensitive products. The facility is equipped for large-scale production, supporting supply chain reliability through inventory management systems and contingency planning. Bulk supply capacity is scalable to accommodate both short-term and long-term orders, ensuring consistent availability for global markets.
Why Choose Us
Pharmaceutical distributors, exporters, and contract manufacturers choose us for our commitment to quality, compliance, and scalability. Our GMP-certified facility ensures pharmaceutical-grade products that meet international standards, while our regulatory expertise streamlines market entry. We offer customized solutions, from formulation development to packaging, with a focus on reliability and innovation. Our global supply chain and bulk production capabilities ensure competitive pricing and on-time delivery, making us a trusted partner for pharmaceutical buyers seeking high-quality, compliant products.
Bulk Supply Inquiry
For bulk supply inquiries, please contact us at info@delwishealthcare.com. Our team is dedicated to providing tailored solutions for pharmaceutical manufacturers and distributors worldwide.
FAQs
What manufacturing standards does Piloherb Cap comply with?
Piloherb Cap is manufactured under cGMP, ISO 9001, and WHO GMP standards, ensuring compliance with global pharmaceutical regulations.
Can Piloherb Cap be customized for specific markets?
Yes, we offer private label and contract manufacturing services to tailor formulations for regional regulatory requirements and market preferences.
What is the shelf life of Piloherb Cap?
The product has a shelf life of 24 months when stored under recommended conditions, with stability testing conducted to ensure potency and safety.
Does Piloherb Cap require special storage conditions?
The Alu Alu packaging provides protection against moisture and light, allowing standard storage conditions.
What quality testing is performed on Piloherb Cap?
Each batch undergoes identity, purity, potency, and dissolution testing, with microbial and heavy metal analysis to ensure compliance with pharmacopeial standards.
Can Piloherb Cap be produced in bulk for export?
Yes, our facility supports large-scale production and bulk supply, with export capabilities to over 50 countries.
What certifications does your facility hold?
Our facility is certified under cGMP, ISO 9001, and ISO 14001, ensuring adherence to international quality and environmental standards.
How do you ensure supply chain reliability?
We maintain a robust logistics network, with inventory management systems and contingency planning to ensure timely delivery and supply chain continuity.
Can I request a sample for quality evaluation?
Yes, we offer sample packs for evaluation, with documentation supporting compliance with pharmacopeial standards.
What is the minimum order quantity for bulk supply?
Minimum order quantities are flexible and tailored to client requirements, with options for both small-scale and large-volume orders.